| |
16:30
|
0227.
 |
Multi-Center and Multi-Vendor Study of Long-TE 1H MRS at
3T for Detection of 2-Hydroxyglutarate in Brain Tumors In Vivo 
Changho Choi1, Thomas Huber2, Anna
Tietze3, Byung Se Choi4, Jung Hee Lee5,
Seung-Koo Lee6, Alexander Lin7, and
Sunitha Thakur8
1UT Southwestern Medical Center, Dallas, TX,
United States, 2Technical
University of Munich, Munich, Germany, 3Aarhus
University Hospital, Aarhus, Denmark, 4Seoul
National University College of Medicine, Seongnam, Korea,
Republic of, 5Sungkyunkwan
University School of Medicine, Seoul, Korea, Republic of, 6Yonsei
University College of Medicine, Seoul, Korea, Republic of, 7Harvard
Medical School, Boston, MA, United States, 8Memorial
Sloan-Kettering Cancer Center, New York, NY, United States
The non-invasive identification of elevated
2-hydroxyglutarate (2HG) in IDH-mutated gliomas by 1H MRS in
vivo is a major breakthrough in brain tumor research.
Studies have shown that optimized long-TE approaches may
confer advantages over short-TE MRS for detecting 2HG. Here
we report an evaluation of the feasibility of long-TE 2HG
MRS in Philips, Siemens and GE 3T scanners. Echo times were
optimized, with numerical simulations and phantom
validation, for the vendor-specific RF pulses. In-vivo data
from IDH-mutated glioma patients, obtained in the three
vendors, are discussed.
|
| |
16:42
 |
0228.
 |
Metabolic Profiling of Malignant Transformation and IDH-mutation
in Diffuse Infiltrating Gliomas 
Llewellyn Jalbert1, Adam Elkhaled1,
Joanna J Phillips2, Evan Neill3,
Marram P Olson3, Mitchel S Berger4,
John Kurhanewicz1,3, Susan M Chang4,
and Sarah J Nelson1,3
1Department of Bioengineering & Therapeutic
Sciences, University of California, San Francisco (UCSF),
San Francisco, CA, United States, 2Department
of Pathology, University of California, San Francisco (UCSF),
San Francisco, CA, United States, 3Department
of Radiology & Biomedical Imaging, University of California,
San Francisco (UCSF), San Francisco, CA, United States, 4Department
of Neurological Surgery, University of California, San
Francisco (UCSF), San Francisco, CA, United States
Patients diagnosed with infiltrating low-grade glioma have a
relatively long survival, and a balance is often struck
between treating the tumor and impacting quality of life.
Aggressive treatments are typically reserved for lesions
that have undergoing malignant transformation (MT) to a
higher-grade lesion. Mutations in the isocitrate
dehydrogenase 1 & 2 oncogenes and production of
2-hydroxyglutarate further characterize these tumors and are
associated with improved outcome and treatment sensitivity.
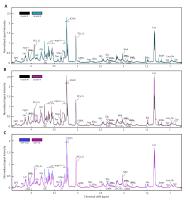
In this study, we found distinct metabolic profiles
associated with patients' tumors that had undergone MT, as
well as contained the IDH-mutated genotype, using proton
HR-MAS spectroscopy.
|
| |
16:54
 |
0229.
 |
Noninvasive Assessment of IDH Mutational Status in Glioma using
MR Elastography 
Kay Pepin1, Arvin Arani1, Mona El
Sheikh1, Nikoo Fattahi1, David Lake1,
Armando Manduca1, Kiaran McGee1, Ian
Parney1, Richard Ehman1, and John
Huston1
1Mayo Clinic, Rochester, MN, United States
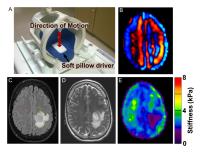
MR elastography (MRE) has been used to characterize the
mechanical properties of normal and diseased brain tissue
(1-4). This study evaluated MRE for the noninvasive
characterization of gliomas, specifically investigating the
relationship between tumor stiffness and mutations in the
IDH1 gene, an important prognostic biomarker for improved
outcome. Eighteen patients were enrolled in this study. MRE
examinations were performed at 3T using an EPI-MRE sequence
and 60Hz vibration frequency. Tumor stiffness was quantified
and compared to IDH mutation status, as determined by
histology. Twelve tumors were identified as IDH1 mutation
positive and were significantly stiffer than tumors with
non-mutated IDH1.
|
| |
17:06
 |
0230.
 |
Amide-Proton-Transfer-Weighted (APTw) MRI as a Surrogate
Biomarker to Detect Recurrent High-grade Gliomas after Treatment
with Chemoradiation: Validation by Image-Guided Stereotactic
Biopsy 
Shanshan Jiang1,2, Charles Eberhart3,
Jaishri Blakeley4, Lindsay Blair4,
Huamin Qin 3,
Michael Lim5, Alfredo Quinones-Hinojosa5,
Hye-Young Heo1, Yi Zhang1, Dong-Hoon
Lee1, Xuna Zhao1, Zhibo Wen2,
Peter C.M. van Zijl1, and Jinyuan Zhou1
1Department of Radiology, Johns Hopkins
University, Baltimore, MD, United States, 2Department
of Radiology, Southern Medical University Zhujiang Hospital,
Guangzhou, China, People's Republic of,3Department
of Pathology, Johns Hopkins University, Baltimore, MD,
United States, 4Department
of Neurology, Johns Hopkins University, Baltimore, MD,
United States, 5Department
of Neurosurgery, Johns Hopkins University, Baltimore, MD,
United States
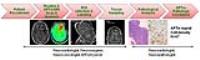
We explored the imaging features of treatment effects and
active tumor in glioma patients after surgery and
chemoradiation using amide-proton-transfer-weighted (APTw)
imaging at 3 Tesla. Needle biopsy samples were obtained for
pathological validation. Corresponding APTw signal
intensities were recorded. Results showed that APTw signal
intensities had strong positive correlations with
cellularity and proliferation. The active tumor had
significantly higher APTw signal intensity, compared to
treatment effects. The area-under-curve (AUC) for APTw
intensities to differentiate treatment effects from active
tumor was 0.959. APT imaging has potential for molecular
image-guided biopsy for post-treatment glioma patients to
distinguish pseudoprogression from tumor recurrence.
|
| |
17:18
|
0231.
 |
Amide Proton Transfer (APT) Imaging of Brain Tumors using 3D
Fast Spin-Echo Dixon Method: Comparison with Separate B0 Mapping 
Osamu Togao1, Akio Hiwatashi1, Jochen
Keupp2, Koji Yamashita1, Kazufumi
Kikuchi1, Masami Yoneyama3, and
Hiroshi Honda1
1Department of Clinical Radiology, Graduate
School of Medical Sciences, Kyushu University, Fukuoka,
Japan, 2Philips
Research, Hamburg, Germany, 3Philips
Electronics Japan, Tokyo, Japan
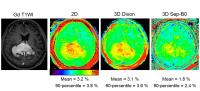
Recently, the FSE Dixon APT acquisition protocol with
intrinsic ?B0 correction was developed and implemented on 3T
clinical MRI scanners. This technique allows simultaneous
acquisition of APT imaging and intrinsic B0 mapping without
increasing scan time. In the present study, we demonstrated
the quantitative performance of the 3D FSE Dixon APT imaging
of brain tumors in comparison with the separate B0 mapping
method.
|
| |
17:30
|
0232.
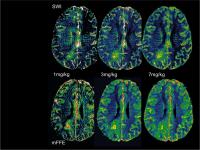 |
Introducing steady state blood volume mapping using ferumoxytol,
a new MRI tool to assess the intravascular space in brain tumors
and other intracranial pathologies 
Csanad Varallyay1, Daniel Schwartz2,
Joao Prola Netto1, Prakash Ambady2,
Andrea Horvath2, and Edward Neuwelt2
1Diagnostic Radiology and Neurology, Oregon
Health and Science University, Portland, OR, United States, 2Neurology,
Oregon Health and Science University, Portland, OR, United
States
Steady state blood volume (SS-CBV) mapping using the blood
pool agent ferumoxytol as an MRI contrast agent is feasible
in brain tumors and other intracranial pathologies. It
allows high resolution, distortion free blood volume maps,
which can be a useful MRI tool to improve diagnosis and
assessment of response to therapy. Ferumoxytol dose and MRI
sequences may be optimized for various clinical
applications.
|
| |
17:42
|
0233.
 |
Semi-quantitative MRI Assessment of anti-PD1 Immunotherapy
Response in Recurrent Glioblastoma 
Lei Qin1,2, Xiang Li2,3, Amanda
Stroiney4, David A Reardon1,2, and
Geoffrey Young2,3
1Dana-Farber Cancer Institute, boston, MA, United
States, 2Harvard
Medical School, boston, MA, United States, 3Brigham
and Women's Hospital, Boston, MA, United States, 4Northeastern
University, Boston, MA, United States
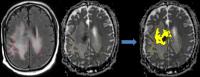
The purpose of this study is to evaluate the predictive
value of quantitative and semi-quantitative MRI biomarkers
in determining patient benefit in anti-PD1 immunotherapy
treatments. Longitudinal MRIs were performed on patients
diagnosed with recurrent GBM. Volumetric analysis of
abnormal tissue from contrast enhanced T1, FLAIR, and ADC
revealed two distinct patterns: a) progressive increase
volume in patients who derived no significant benefit, and
b) a transient increase in the volume, followed by a delayed
decrease in patients with >6 mo survival on trial. In this
preliminary study (n=10), the data suggest that the volume
of abnormal tissue on ADC seems to correlate better with
patient benefit than abnormality on FLAIR and T1.
|
| |
17:54
|
0234.
 |
Serial 3D H-1 MRSI of Patients with Newly Diagnosed GBM being
Treated with Radiation, Temozolomide, Erlotinib and Bevacizumab 
Sarah Nelson1, Yan Li1, Janine Lupo1,
Marram Olson1, Jason Crane1, Annette
Molinaro2, Ritu Roy3, Soonmee Cha1,
and Susan Chang2
1Radiology and Biomedical Imaging, University of
California, San Francisco, San Francisco, CA, United States, 2Neurological
Surgery, University of California, San Francisco, San
Francisco, CA, United States, 3Helen
Diller Family Comprehensive Cancer Center, University of
California, San Francisco, San Francisco, CA, United States
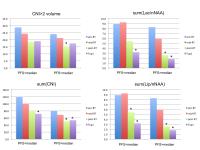
Patients with newly diagnosed GBM are typically treated with
a combination of radiation and temozolomide in conjunction
with a variety of investigational agents. Assessing the
effectiveness of such therapies is complicated by
differences in their mechanisms of action that lead to
ambiguities in the interpretation of conventional anatomic
images and difficulties in assessing the spatial extent of
tumor. The results of this study demonstrate that
integrating 3D lactate edited H-1 MRSI into routine MR
examinations and applying quantitative analysis methods
allows for the objective evaluation of changes in tumor
burden and the early assessment of outcome.
|
| |
18:06
|
0235.
 |
Differential imaging biomarker response to sunitinib across
tumor histologies in a prospective trial of brain metastases 
Caroline Chung1, Brandon Driscoll1,
Warren Foltz1, Cynthia Menard1, David
Jaffray1, and Catherine Coolens1
1Princess Margaret Cancer Centre, Toronto, ON,
Canada
Our preclinical study of sunitinib (SU) in combination with
conformal large single fraction radiation in an orthotopic
murine brain tumor model, discovered that changes in
apparent diffusion coefficient (ADC), AUC and Ktrans were
promising imaging biomarkers that could predict response to
SU as well as combined SU and radiation. Based on our
preclinical findings, we designed a prospective phase I
trial of SU and radiosurgery (SRS) for brain metastases that
incorporated translational investigation of these imaging
biomarkers. Here we summarize our discovery of differential
ADC and AUC responses to sunitinib between renal cell cancer
and other histology brain metastases.
|
| |
18:18
 |
0236.
 |
Optimal time-window and perfusion protocol for MRI in early
assessment of high grade glioma treatment response 
Christopher Larsson1,2, Jonas Vardal1,
Inge Rasmus Groote3, Magne Mørk Kleppestø1,2,
Petter Brandal4, and Atle Bjørnerud1,5
1The Intervention Centre, Oslo University
Hospital, Oslo, Norway, 2Faculty
of Medicine, University of Oslo, Oslo, Norway, 3Department
of Psychology, University of Oslo, Oslo, Norway, 4Department
of Cancer Medicine, Surgery & Transplantation, Oslo
University Hospital, Oslo, Norway, 5Faculty
of Physics, University of Oslo, Oslo, Norway
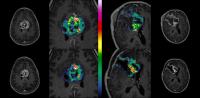
Due to limitations in structural MRI in assessment of
overall survival (OS) in high grade glioma interest in more
advanced functional MRI methods has risen. A prospective
longitudinal high grade glioma study including structural
imaging and T1/T2* perfusion was performed in 27 patients to
investigate the optimal time-window and most sensitive MRI
perfusion method for early OS analysis.
No structural imaging, DSC or
absolute perfusion parameter was found significant for early
OS assessment. Change in median Ktrans and CBF from baseline
to eight weeks was found significant and CBF change >15%
most accurate predictor for poor OS. |
|











