| |
13:30
 |
0656.
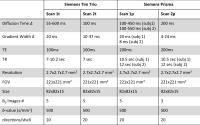 |
What dominates the diffusivity time dependence transverse to
axons: Intra- or extra-axonal water? 
Hong-Hsi Lee1, Jelle Veraart1,2,
Dmitry S. Novikov1, and Els Fieremans1
1New York University, Center for Biomedical
Imaging, New York, NY, United States, 2iMinds-Vision
Lab, University of Antwerp, Antwerp, Belgium
Diffusion MRI enables to evaluate microstructure at the
mesoscopic scale. In particular, tuning the diffusion time
over a wide range could increase the sensitivity for
acquiring useful biomarkers, such as the axonal diameter or
density. However, it is unclear whether either intra-, or
extra-axonal water attribute most to the observed changes of
diffusion signal with diffusion time. Here, we evaluate the
dependence of the diffusion coefficient (obtained from the
diffusion signal at low $$$b$$$-value) on $$$\delta$$$ and
$$$\Delta$$$ in the direction perpendicular to axons in the
human brain, and explain these dependencies by diffusion of
water in the extra-axonal space.
|
| |
13:42
|
0657.
 |
Impact of transcytolemmal water exchange on quantitative
characterization of tissue microstructure using diffusion MRI 
Hua Li1, Xiaoyu Jiang1, Jingping Xie1,
John C Gore1, and Junzhong Xu1
1Radiology and Radiological Sciences, Vanderbilt
University, Nashville, TN, United States
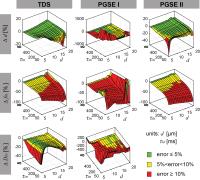
Current diffusion MRI methods that quantitatively
characterize tissue microstructure usually assume a zero
transcytolemmal water exchange rate $$$\tau_{in}$$$ between
intra- and extracellular spaces. This assumption may not be
true in many cases of interest. The present work used both
computer simulations and cell culture in vitro to
investigate the influence of $$$\tau_{in}$$$ on the accuracy
of fitted microstructural parameters such as mean cell size
$$$d$$$, intracellular water fraction $$$f_{in}$$$ and
diffusion coefficient $$$D_{in}$$$. Results indicate $$$d$$$
is relatively insensitive to $$$\tau_{in}$$$, while $$$f_{in}$$$
is always underestimated with finite $$$\tau_{in}$$$. $$$D_{in}$$$
can be fit reliably only when short diffusion times are
used.
|
| |
13:54
 |
0658.
 |
Low frequency oscillating gradient spin-echo sequences improves
sensitivity to axon diameter – an experimental validation study
in live nerve tissue 
Lebina Shrestha Kakkar1, Oscar Bennett1,
David Atkinson2, Bernard Siow3, James
Phillips4, Simon Richardson3, Enrico
Kaden1, and Ivana Drobnjak1
1Centre for Medical Image Computing, University
College London, London, United Kingdom, 2Centre
for Medical Imaging, University College London, London,
United Kingdom, 3Centre
for Advanced Biomedical Imaging, University College London,
London, United Kingdom, 4Department
of Biomaterials & Tissue Engineering, University College
London, London, United Kingdom
In a recent simulation study, Drobnjak et
al demonstrates
that low-frequency oscillating gradient spin-echo (OGSE)
sequence is more sensitive to axon diameter than
conventional pulsed gradient spin-echo (PGSE) sequence when
fibre orientation is unknown or when fibre dispersion
exists. Here, we experimentally validate this claim. We
image a live rat sciatic nerve tissue using both sequences
and compare its agreement with histology. Our results
confirm that OGSE provides more accurate and precise
diameter estimates compared to PGSE. Additionally, OGSE
parameter estimates are less affected by reduced number of
diffusion gradient directions, suggesting their use could
translate into faster scan times.
|
| |
14:06
|
0659.
 |
Asymmetries of the dendrite density in cortical areas assessed
by diffusion MR microscopy using NODDI 
Achille Teillac1,2,3, Sandrine Lefranc2,3,4,
Edouard Duchesnay2,3,4,5, Fabrice Poupon2,3,4,
Maite Alaitz Ripoll Fuster1,2,3, Denis Le Bihan1,2,3,
Jean François Mangin2,3,4,5, and Cyril Poupon1,2,3,5
1CEA NeuroSpin / UNIRS, Gif-sur-Yvette, France, 2Université
Paris-Saclay, Orsay, France, 3France
Life Imaging, Orsay, France, 4CEA
NeuroSpin / UNATI, Gif-sur-Yvette, France, 5http://cati-neuroimaging.com/,
Gif-sur-Yvette, France
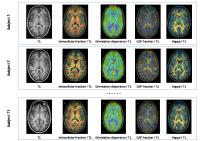
In this study, we investigated the dendrite density in
cortical areas with diffusion MR microscopy using the NODDI
model and showed, on a population of healthy volunteers,
significant differences between left and right hemisphere,
correlated with their supported brain functions.
|
| |
14:18
|
0660.
 |
Diffusion-Relaxation Correlation Spectroscopic Imaging (DR-CSI):
An Enhanced Approach to Imaging Microstructure 
Daeun Kim1, Joong Hee Kim2, and Justin
P Haldar1
1Department of Electrical Engineering, University
of Southern California, Los Angeles, CA, United States, 2Department
of Neurology, Washington University, St. Louis, MO, United
States
We propose a new MR experiment called diffusion-relaxation
correlation spectroscopic imaging (DR-CSI). DR-CSI acquires
imaging data across a range of different b-value and echo
time combinations, and then performs regularized
reconstruction to generate a 2D diffusion-relaxation
correlation spectrum for every voxel. The peaks of this
spectrum correspond to the different tissue
microenvironments that are present within each macroscopic
imaging voxel, which provides powerful insight into the
tissue microstructure. Compared to standard relaxometry or
diffusion imaging, DR-CSI provides unique capabilities to
resolve tissue compartments that have similar relaxation or
diffusion parameters. DR-CSI is demonstrated with spinal
cord traumatic injury MRI data.
|
| |
14:30
|
0661.
 |
Linear Multi-scale Modeling of diffusion MRI data: A framework
for characterization of orientational structures across length
scales 
Barbara Wichtmann1,2, Susie Huang1,
Qiuyun Fan1, Thomas Witzel1, Elizabeth
Gerstner3, Bruce Rosen1, Lothar Schad2,
Lawrence Wald1,4, and Aapo Nummenmaa1
1A. A. Martinos Center for Biomedical Imaging,
Department of Radiology, Massachusetts General Hospital,
Charlestown, MA, United States, 2Computer
Assisted Clinical Medicine, Medical Faculty Mannheim,
Heidelberg University, Mannheim, Germany, 3Department
of Neurology, Massachusetts General Hospital, Harvard
Medical School, Boston, MA, United States, 4Harvard-MIT
Division of Health Sciences and Technology, Massachusetts
Institute of Technology, Cambridge, MA, United States
We propose a new analysis technique called Linear
Multi-scale Modeling (LMM) for diffusion MRI data that
enables detailed microstructural tissue characterization by
separating orientation distributions of restricted and
hindered diffusion water compartments over a range of length
scales. We demonstrate the ability of LMM to estimate volume
fractions, compartment sizes and orientation distributions
utilizing both simulations as well as empirical data from
one healthy subject and one tumor patient acquired using a
human 3T MRI scanner equipped with a 300mT/m gradient
system. Possible applications of our modeling framework
include characterization of diffusion microstructural
signatures of pathological vs. healthy tissue.
|
| |
14:42
|
0662.
 |
Microscopic Interpretation and Generalization of the
Bloch-Torrey Equations for Diffusion MR 
Inbar Seroussi1, Ofer Pasternak1,2,
and Nir Sochen 1
1Tel-Aviv university, Tel Aviv, Israel, 2Psychiatry
and Radiology, Harvard Medical School, Boston, MA, United
States
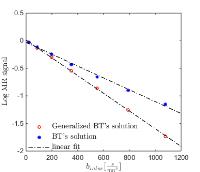
How to bridge microscopic molecular motion with macroscopic
diffusion MR signal? We suggest a simple stochastic
microscopic model for molecular motion within a magnetic
field. We derive the Fokker-Planck equation of this model,
which is an analytic expression of the probability density
function describing the magnetic diffusion propagator. This
propagator is a crucial quantity and provides the link
between the microscopic equations and the measured MR
signal. Using the propagator we derive a generalized version
for the macroscopic Bloch-Torrey equation. The advantage of
this derivation is that it does not require assumptions such
as constant diffusion coefficient, or ad-hoc selection of a
propagator. In fact, we show that the generalized
Bloch-Torrey equations have an additional term that was
previously neglected and accounts for spatial varying
diffusion coefficient. Including this term better predicts
MR signal in complex microstructures, such as those expected
in most biological experiments.
|
| |
14:54
|
0663.
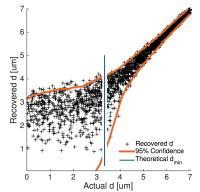 |
Estimating the axon diameter from intra-axonal water diffusion
with arbitrary gradient waveforms: Resolution limit in parallel
and dispersed fibers 
Markus Nilsson1, Samo Lasic2, Daniel
Topgaard3, and Carl-Fredrik Westin4
1Lund University Bioimaging Center, Lund
University, Lund, Sweden, 2CR
Development AB, Lund, Sweden, 3Physical
Chemistry, Lund University, Lund, Sweden, 4Brigham
and Women's Hospital, Harvard Medical School, Boston, MA,
United States
The use of non-conventional gradient waveforms has brought
renewed interest in non-invasive estimation of the axon
diameter from diffusion MRI. Using conventional single
diffusion encoding (SDE) at clinical MRI scanners, axons
smaller than the resolution limit (4-5 microns) were
indistinguishable from each other. We here predict the
resolution limit for arbitrary gradient waveforms in systems
with (i) parallel axons and (ii) orientation dispersion.
Results show that SDE is optimal for parallel fibers, but
that multiple diffusion encodings (MDE) are preferred where
there is orientation dispersion. With 300 mT/m and MDE, the
resolution limit was 2.8 microns for dispersed fibers.
|
| |
15:06
|
0664.
 |
Quantification of anisotropy and directionality in
three-dimensional electron microscopy images and diffusion
tensor imaging of injured rat brain 
Raimo A. Salo1, Ilya Belevich2, Eppu
Manninen1, Eija Jokitalo2, Olli Gröhn1,
and Alejandra Sierra1
1Department of Neurobiology, A. I. Virtanen
Institute for Molecular Sciences, University of Eastern
Finland, Kuopio, Finland, 2Institute
of Biotechnology, University of Helsinki, Helsinki, Finland
Diffusion tensor imaging (DTI) is a widely used tool,
however, the contribution of brain tissue microstructure
into DTI contrast is not fully understood. In this work, we
propose using serial block-face scanning electron microscopy
(SBEM) and Fourier analysis to gain insight into this
contribution. We calculated anisotropy and orientation from
SBEM stacks and compare the values to fractional anisotropy
and orientation from in
vivo and ex
vivo DTI.
This work will give new insights to the contribution of
microstructure to DTI contrast in normal brain and during
pathology.
|
| |
15:18
|
0665.
 |
A new paradigm to assess brain cell microstructure by
diffusion-weighted magnetic resonance spectroscopy: proof of
concept and initial results in the macaque brain 
Marco Palombo1,2, Clémence Ligneul1,2,
Chloé Najac1,2, Juliette Le Douce1,2,
Julien Flament1,2, Carole Escartin1,2,
Philippe Hantraye1,2, Emmanuel Brouillet1,2,
Gilles Bonvento1,2, and Julien Valette1,2
1CEA/DSV/I2BM/MIRCen, Fontenay-aux-Roses, France, 2CNRS
Université Paris-Saclay UMR 9199, Fontenay-aux-Roses, France
We introduce a novel paradigm for non-invasive brain
microstructure quantification, where original diffusion
modeling is merged with cutting-edge diffusion-weighted
spectroscopy (DW-MRS) experiments to capture features of
cellular morphology that have remained largely ignored by
DW-MRI. A compact description of long-range cellular
morphology is used to randomly generate large collections
of synthetic cells where particles diffusion is simulated.
After investigating model robustness, we apply it on
metabolite ADC measured in vivo in the monkey brain up to td=2
seconds. The new paradigm introduced here opens new
possibilities to non-invasively extract quantitative
information about cell size, complexity and heterogeneity in
the brain.
|
|












