| |
10:00
|
|
Introduction |
| |
10:12
 |
0288.
 |
Radiogenomic analysis of glioblastoma using protein-based amide
proton transfer (APT) imaging and message RNA expression: A
novel correlation in molecular imaging and gene characteristics 
Shanshan Jiang1, Xianlong Wang1, Hao
Yu1, Jiandong Xi1, Jingwen Wu1,
Lisong Liang1, Shilong Lu1, Tianyu Zou1,
Jinyuan Zhou2, and Zhibo Wen1
1Department of Radiology, Southern Medical
University Zhujiang Hospital, Guangzhou, China, People's
Republic of, 2Department
of Radiology, Johns Hopkins University, Baltimore, MD,
United States
The correlation between endogenous protein-based
APT-weighted (APTw) imaging and gene expression in
glioblastoma (GBM) was investigated. 16 patients with newly
diagnosed GBM were studied. APTw/FLAIR hyperintensity area
ratio (AFR), and APTw hyperintensity/gadolinium
contrast-enhanced T1w enhancement area ratio (ATR) were
calculated. Interoperative paired tumor and adjacent normal
tissues were sampled for genomic analysis. BRCA1 and CDK4
were significantly downregulated in the high AFR group
(adjusted P= 0.000953 and 0.025187), and SLAMF9 and MIA were
significantly downregulated in the high ATR group (adjusted
P= 1.08E-11 and 0.00997). APT imaging has great potential
for unveiling some special genomic changes in GBM.
|
| |
10:24
|
0289.
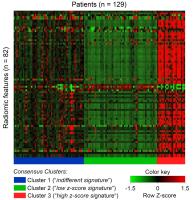 |
Large-scale radiomic profiling of glioblastoma identifies an
imaging signature for predicting and stratifying antiangiogenic
treatment response. 
Philipp Kickingereder1, Michael Götz2,
John Muschelli3, Antje Wick4, Ulf
Neuberger5, Russell T Shinohara6,
Alexander Radbruch7, Heinz-Peter Schlemmer7,
Wolfgang Wick4, Martin Bendszus5,
Klaus H Maier-Hein2, and David Bonekamp7
1Department of Neuroradiology, University
Hospital Heidelberg, Heidelberg, Germany, 2Division
Medical and Biological Informatics, DKFZ - German Cancer
Research Center, Heidelberg, Germany, 3Department
of Biostatistics, Johns Hopkins Bloomberg School of Public
Health, Baltimore, MD, United States, 4Department
of Neurology, University of Heidelberg Medical Center,
Heidelberg, Germany, 5Department
of Neuroradiology, University of Heidelberg Medical Center,
Heidelberg, Germany, 6Department
of Biostatistics and Epidemiology, Perelman School of
Medicine, University of Pennsylvania, Philadelphia, PA,
United States, 7Department
of Radiology, DKFZ - German Cancer Research Center,
Heidelberg, Germany
To analyze the potential of radiomics, an emerging field of
research that aims to utilize the full potential of medical
Imaging (1,2), for predicting and stratifying treatment
response to antiangiogenic therapy in patients with
recurrent glioblastoma.
|
| |
10:36
|
0290.
 |
Low Apparent Diffusion Coefficient Values Correlate with
Enhancing Mitosis and Cell Proliferation Expression in
glioblastoma using Locus-Specific Radiogenomic Map - Permission Withheld
Cheng-Yu Chen1,2,3, Fei-Ting Hsu1,
Hua-Shan Liu1,4, Ping-Huei Tsai1,3,
Chia-Feng Lu2,3,5, Yu-Chieh Kao2,3,
Li-Chun Hsieh1, and Pen-Yuan Liao1
1Department of Medical Image, Taipei Medical
University Hospital, Taipei, Taiwan, 2Translational
Imaging Research Center, College of Medicine, Taipei Medical
University, Taipei, Taiwan, 3Department
of Radiology, School of Medicine, Taipei Medical University,
Taipei, Taiwan, 4Graduate
Institute of Clinical Medicine, Taipei Medical University,
Taipei, Taiwan, 5Department
of Biomedical Imaging and Radiological Sciences, National
Yang-Ming University, Taipei, Taiwan
A new approach to unravel the genomic expression of
glioblastoma by advanced MR imaging technique has recently
been introduced to improve the prognostic and predictive
efficacies of neuroimaging. This imaging method is
potentially a valuable tool to link individual differences
in the human genome to structure, function and physiology
into brain disease, a method referred to as radiogenomics.
In this study, we established locus specific radiogenomic
map based on MR imaging and Microarray RNA analysis. Our
results revealed that apparent diffusion coefficient (ADC)
differences were correlated with several biological
processes change, including cell proliferation, T cell
immunity, immune response, and mitosis. The identification
of tumor genotypes by imaging phenotypes will open a new era
of therapeutic strategy in high grade gliomas.
|
| |
10:48
|
0291.
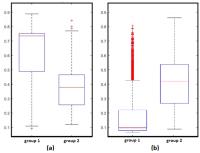 |
Radiomic features on Multi-parametric MRI can help risk
categorization of Prostate Cancer patients on Active
Surveillance 
Ahmad Algohary1, Satish Viswanath1,
Sadhna Verma2, and Anant Madabhushi1
1Case Western Reserve University, Cleveland, OH,
United States, 2University
of Cincinnati, Cincinnati, OH, United States
Active Surveillance (AS) offers an important alternative to
radical treatment as more men die with prostate cancer (PCA)
than of the disease. In this study, we explore the role of
radiomic texture features on a pre-biopsy screening 3 Tesla
multi-parametric MRI that can predict which men with
elevated PSA will have a cancer-positive or cancer-negative
biopsy. The selected texture features correctly identified
14/15 biopsy-negative (compared to 10/15 cases correctly
identified by PIRADS) and 23/30 biopsy-positive cases
(compared to only 15/30 correctly identified by PIRADS).
These features appear to enhance differentiation between
biopsy-positive and biopsy-negative prostate cancer patients
on Active Surveillance.
|
| |
11:00
|
0292.
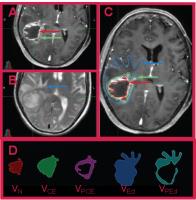 |
Association of Radiomics and Metabolic Tumor Volumes in
Radiation Treatment of Glioblastoma Multiforme - Permission Withheld
christopher lopez1, Natalya Nagornaya2,
Nestor Parra2, Deukwoo Kwon2, Fazilat
Ishkanian2, Arnold Markoe2, Andrew
Maudsley2, and Radka Stoyanova2
1Radiation Oncology, University of Miami, Miami,
FL, United States, 2University
of Miami, Miami, FL, United States
To investigate the importance of metabolites of N-acetyl
aspartate and choline derived from MRSI and the correlation
of image features from localized radiation therapy volumes
determined from MRI and CT defined tumor volumes. Also to
replace subjective categorical image features with
calculated objective features. Results suggest that
radiation therapy planning can be more accurate by adding
metabolic information.
|
| |
11:12
|
0293.
 |
Relationship of invivo MR parameters to molecular
characteristics of non-enhancing lower-grade gliomas 
Tracy L Luks1, Tracy Richmond McKnight1,
Aurelia Williams1, Evan Neill1,
Khadjia Lobo1, Anders Persson2, Arie
Perry3, Joanna Phillips3, Annette
Molinaro4, Susan Chang4, and Sarah J
Nelson1
1Radiology and Biomedical Imaging, University of
California San Francisco, San Francisco, CA, United States, 2Neurology,
University of California San Francisco, San Francisco, CA,
United States, 3Pathology,
University of California San Francisco, San Francisco, CA,
United States, 4Neurosurgery,
University of California San Francisco, San Francisco, CA,
United States
Invivo MR anatomy, diffusion, perfusion, and spectroscopy
profiles from non-enhancing grade 2 and grade 3 gliomas were
examined by histologic and molecular characteristics
associated with clinical outcome. Patients underwent a
pre-surgical 3T MR exam including IRSPGR, FSE, FLAIR, DWI,
MRSI and DSC. For surgical biopsies, histological sub-type,
grade, cleaved caspase-3, MIB-1, Ki67, IDH1R132H, ATRX, p53,
and co-deletion of 1p19q were determined. Overall, molecular
characteristics associated with worse clinical outcome were
associated with higher ADC and lower FA, lower nCBV and nPH,
and higher Recov, and higher nLAC.
|
| |
11:24
|
0294.
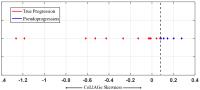 |
Radiomic features from the necrotic region on post-treatment
Gadolinium T1w MRI appear to differentiate pseudo-progression
from true tumor progression in primary brain tumors 
Prateek Prasanna1, Raymond Huang2,
Andrew Rose1, Gagandeep Singh1, Anant
Madabhushi1, and Pallavi Tiwari1
1Case Western Reserve University, Cleveland, OH,
United States, 2Brigham
and Women's Hospital, Boston, MA, United States
Pseudoprogression is an early-delayed inflammatory response
to chemoradiotherapy typically appearing up to 3 months
post-treatment in brain tumors. On routine MRI,
pseudoprogression closely mimics the appearance of true
progression, thereby making their visual identification
challenging. Early diagnosis of pseudoprogression has
implications in management of treatment effects and
subsequently survival. We present initial results of using a
newly developed radiomic descriptor, CoLlAGE, in
distinguishing the two pathologies. We report that CoLlAGe
measurements when captured from the necrotic region as
opposed to just the enhancing region on MRI can reliably
distinguish psuedo-progression from true progression
with 100% accuracy (n=17)
|
| |
11:36
|
0295.
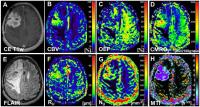 |
Combined assessment of tumor oxygen metabolism and angiogenesis
in glioma patients 
Andreas Stadlbauer1, Max Zimmermann1,
Karl Rössler1, Stefan Oberndorfer2,
Arnd Dörfler3, Michael Buchfelder1,
and Gertraud Heinz4
1Department of Neurosurgery, University of
Erlangen, Erlangen, Germany, 2Department
of Neurology, University Clinic of St. Pölten, St. Pölten,
Austria, 3Department
of Neuroradiology, University of Erlangen, Erlangen,
Germany, 4Department
of Radiology, University Clinic of St. Pölten, St. Pölten,
Austria
Reprogramming energy metabolism and inducing angiogenesis
are part of the hallmarks of cancer. Thirty-five patients
with untreated or recurrent glioma were examined using
vascular architecture mapping (VAM) and the multiparametric
quantitative BOLD (mp-qBOLD) technique for combined
exanimation of oxygen metabolism and angiogenesis in gliomas.
Maps of oxygen extraction fraction (OEF) and cerebral
metabolic rate of oxygen (CMRO2) as well as of
the vascular architecture MRI biomarkers microvessel radius
(RU), density (NU), and type indicator
(MTI) were calculated. Low-grade glioma showed increased OEF.
Glioblastomas showed significantly increased CMRO2 and
NU. MTI demonstrated widespread areas draining
venous microvasculature in high-grade gliomas.
|
| |
11:48
|
0296.
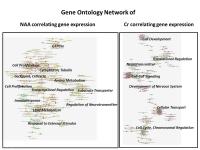 |
Radiogenomics by Proton Magnetic Resonance Spectroscopy:
Integrative Analysis of Metabolites and Genome-wide Expression
in Glioblastomas 
Dieter Henrik Heiland1, Thomas Lange2,
Ralf Schwarzwald3, Dietmar Pfeifer4,
Karl Egger3, Horst Urbach3, Astrid
Weyerbrock1, and Irina Mader3
1Dept. of Neurosurgery, University Medical Center
Freiburg, Freiburg, Germany, 2Dept.
MR Physics, Dept. of Radiology, University Medical Center
Freiburg, Freiburg, Germany, 3Dept.
of Neuroradiology, University Medical Center Freiburg,
Freiburg, Germany, 4Department
of Hematology, Oncology and Stem Cell Transplantation,
University Medical Center Freiburg, Freiburg, Germany
The purpose of this work was to search for a connection
between metabolites observed by proton magnetic resonance
spectroscopy of glioblastomas and tumor genetics. Two
specific pathways could be identified, one belonging to NAA
and discriminating an astroglial versus oligo/neural
subgroup. Another one was related to Cr also distinguishing
between two subgroups, one attributed to apoptosis and
another one to the PI3K-AKT-mTOR signaling cascade.
|
|








