Digital Posters
CEST
ISMRM & SMRT Annual Meeting • 15-20 May 2021

| Concurrent 4 | 17:00 - 18:00 |
1451.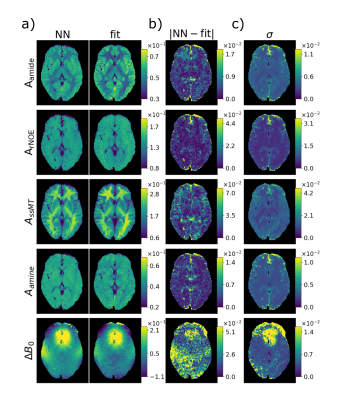 |
DeepCEST: 7T Chemical exchange saturation transfer MRI contrast inferred from 3T data via deep learning with uncertainty quantification
Leonie E. Hunger1, Alexander German1, Felix Glang2, Katrin M. Khakzar1, Nam Dang1, Angelika Mennecke1, Andreas Maier3, Frederik Laun4, and Moritz Zaiss1,2
1Department of Neuroradiology, University Hospital Erlangen, Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU), Erlangen, Germany, 2High-field Magnetic Resonance Center, Max Planck Institute for Biological Cybernetics, Tübingen, Germany, 3Pattern Recognition Lab, Friedrich-Alexander-University Erlangen-Nürnberg, Erlangen, Germany, 4Institute of Radiology, University Hospital Erlangen, Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU), Erlangen, Germany
The deepCEST approach enables to perform CEST experiments at a lower magnetic field strength and predict the contrasts of a higher field strength. This is possible through the application of a neural network, which was trained with low and high B1 Z-spectra acquired at 3T as input data, and as target data 5-pool-Lorentzian fitted amplitudes obtained from 7T spectra were used. The network included an uncertainty quantification to verify the reliability of the predicted images.
|
|||
1452.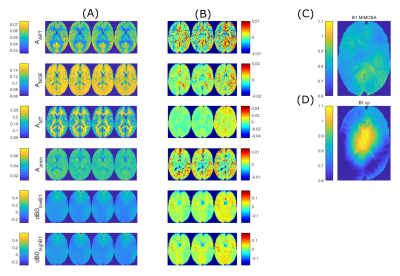 |
Reduction of 7T CEST scan time and evaluation by L1-regularised linear projections
Moritz Simon Fabian1, Felix Glang2, Katrin Michaela Khakzar1, Angelika Barbara Mennecke1, Alexander German1, Manuel Schmidt3, Burkhard Kasper4, Arnd Dörfler1, Frederik B. Laun1, and Moritz Zaiss1
1Department of Neuroradiology, University Hospital Erlangen, Friedrich Alexander University Erlangen-Nürnberg, Erlangen, Germany, 2High-field Magnetic Resonance Center, Max Planck Institute for Biological Cybernetics, Tübungen, Germany, 3Department of Neuroradiology, University Hospital Erlangen, Erlangen, Germany, 4Department of Neurology, Epilepsy Center, University Hospital Erlangen, Erlangen, Germany
Measurement and evaluation of multi-parametric CEST protocols requires complex and time consuming processing for correction of field inhomogeneities and contrast generation. In this work, we expand the linear projection approach for mapping motion corrected 7T CEST data directly to Lorentzian target parameters by L1-regularisation. This translates to subsampling in the frequency offset domain, resulting in reduced acquisition time. The method generalizes from healthy subject training data to unseen healthy test data and a tumor patient dataset. The L1-regularized linear projection approach integrates shortcutting of B0 and B1 correction, denoising, and Lorentzian fitting. It enforces sparsity of required frequency offsets.
|
|||
1453.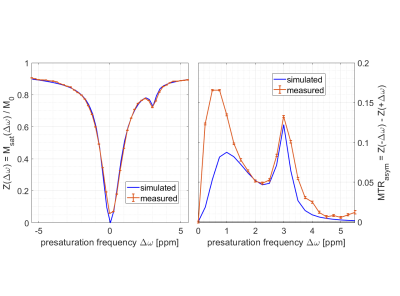 |
Open source Pulseq interpreter for CEST MRI on Bruker systems
Sebastian Mueller1, Kai Herz1, Klaus Scheffler1,2, and Moritz Zaiss1,3
1High-field Magnetic Resonance Center, Max Planck Institute for Biological Cybernetics, Tuebingen, Germany, 2Department of Biomedical Magnetic Resonance, Eberhard Karls University Tuebingen, Tuebingen, Germany, 3Department of Neuroradiology, University Hospital Erlangen, Erlangen, Germany
CEST MRI becomes a frequently applied tool, however, a lot of method development is still done on pre-clinical scanners. Here we propose an approach that allows a fully automatic combination of existing MRI readouts for Bruker systems with open source pulseq-files for definition of CEST pre-saturation, without additional sequence programming. We believe that our work facilitates both basic research on CEST MRI and straightforward implementation of sophisticated, precisely defined CEST experiments on Bruker systems. Additionally, the use of Pulseq provides a universal, sharable standard to run the exact same pre-saturation in simulations and on clinical devises at a later stage.
|
|||
1454. |
Linear projection-based CEST reconstruction – the simplest explainable AI
Felix Glang1, Moritz Fabian2, Alex German2, Katrin Khakzar2, Angelika Mennecke2, Frederik Laun3, Burkhard Kasper4, Manuel Schmidt2, Arnd Doerfler2, Klaus Scheffler1,5, and Moritz Zaiss1,2
1High-field Magnetic Resonance Center, Max Planck Institute for Biological Cybernetics, Tübingen, Germany, 2Department of Neuroradiology, University Hospital Erlangen, Erlangen, Germany, 3Institute of Radiology, University Hospital Erlangen, Erlangen, Germany, 4Neurology, Epilepsy Center, University Clinic of Friedrich Alexander University Erlangen-Nürnberg, Erlangen, Germany, 5Department of Biomedical Magnetic Resonance, Eberhard Karls University Tübingen, Tübingen, Germany
Evaluation of multi-parametric in vivo CEST MRI often requires complex computational processing for both field inhomogeneity correction and contrast generation. In this work, linear regression was used to obtain coefficient vectors that directly map uncorrected 7T spectra to corrected Lorentzian target parameters by simple linear projection. The method generalizes from healthy subject training data to unseen test data of both healthy subjects and tumor patients. The linear projection approach thus integrates correction of both B0 and B1 inhomogeneity as well as contrast generation in a single fast and interpretable computation step.
|
|||
1455.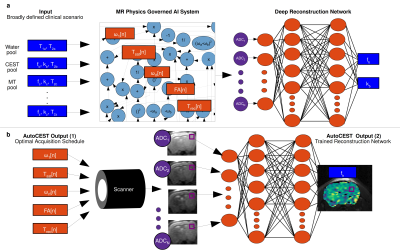 |
In-Vivo Sub-Minute rNOE Mapping Using AutoCEST: a Machine-Learning Approach for CEST/MT Protocol Invention and Quantitative Reconstruction
Or Perlman1, Bo Zhu1,2, Moritz Zaiss3,4, Naoyuki Shono5, Hiroshi Nakashima5, E. Antonio Chiocca5, Matthew S. Rosen1,2, and Christian T. Farrar1
1Athinoula A. Martinos Center for Biomedical Imaging, Department of Radiology, Massachusetts General Hospital and Harvard Medical School, Charlestown, MA, United States, 2Department of Physics, Harvard University, Cambridge, MA, United States, 3Magnetic Resonance Center, Max Planck Institute for Biological Cybernetics, Tübingen, Germany, 4Department of Neuroradiology, University Clinic Erlangen, Erlangen, Germany, 5Brigham and Women’s Hospital and Harvard Medical School, Boston, MA, United States
The long acquisition-time and the semi-quantitative nature of the typical CEST-MRI experiment constitute a major obstacle for its clinical adoption. Recently, a machine-learning approach termed AutoCEST was developed, for the automatic design of the optimal acquisition schedule and the reconstruction of quantitative 2-pool CEST maps. Here, we expand this approach for in-vivo scenarios, by incorporating the semisolid-pool into the underlying computational-graph and allowing 3 pools. AutoCEST was evaluated for quantitative rNOE mapping using a GBM mouse model, resulting in a total acquisition and reconstruction times of 49.15s. The tumor rNOE volume-fraction was significantly decreased, in agreement with previous human studies.
|
|||
1456.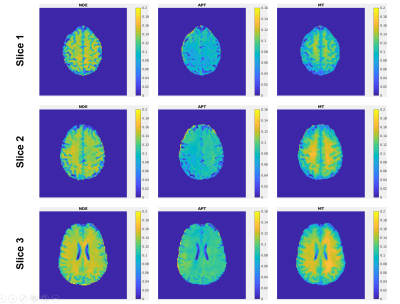 |
Whole-Brain Steady-State CEST at 3T Using MR Multitasking
Pei Han1,2, Karandeep Cheema1,2, Hsu-Lei Lee1, Zhengwei Zhou1, Tianle Cao1,2, Sen Ma1, Nan Wang1, Anthony G. Christodoulou1,2, and Debiao Li1,2
1Biomedical Imaging Research Institute, Cedars-Sinai Medical Center, Los Angeles, CA, United States, 2Department of Bioengineering, UCLA, Los Angeles, CA, United States
We propose a fast 3D steady-state CEST (ss-CEST) method at 3T using MR Multitasking. By exploiting the correlation among images throughout the spatial, time, and offset frequency dimensions, the low-rank tensor framework shows a possibility for at least 2x acceleration of ss- CEST. High-quality quantitative maps can be generated from ss-CEST images. The Z-spectrum acquisition with whole-brain coverage can be done within 5.5min.
|
|||
1457.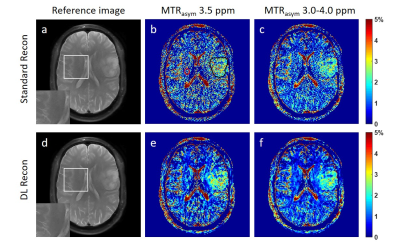 |
Deep Learning-based image reconstruction improves CEST MRI
Shu Zhang1, Xinzeng Wang2, F. William Schuler1, R. Marc Lebel3, Mitsuharu Miyoshi4, Ersin Bayram2, Elena Vinogradov5, Jason Michael Johnson6, Jingfei Ma7, and Mark David Pagel1,7
1Cancer Systems Imaging, MD Anderson Cancer Center, Houston, TX, United States, 2Global MR Applications & Workflow, GE Healthcare, Houston, TX, United States, 3Global MR Applications & Workflow, GE Healthcare, Calgary, AB, Canada, 4Global MR Applications & Workflow, GE Healthcare Japan, Tokyo, Japan, 5Radiology, UT Southwestern Medical Center, Dallas, TX, United States, 6Neuroradiology, MD Anderson Cancer Center, Houston, TX, United States, 7Imaging Physics, MD Anderson Cancer Center, Houston, TX, United States
Chemical exchange saturation transfer (CEST) measurements can be compromised by a low signal-to-noise ratio (SNR) due to the small CEST contrast in vivo. Deep learning-based image reconstruction (DL Recon) can enhance image SNR without losing image resolution or altering the image contrast, hence has the potential to improve quantitative CEST measurements. In this study, we investigated the improvement to CEST quantitation by DL Recon in glioma patients. We found that DL Recon substantially reduced the noise in the MTRasym maps and improved the lesion conspicuity.
|
|||
1458.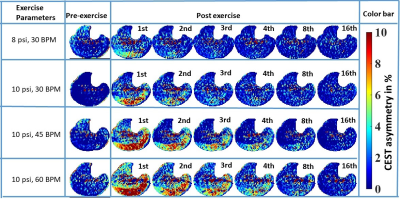 |
Indirect Inference of Acidification in Exercised Skeletal Muscle using Creatine CEST
Dushyant Kumar1, Ryan Armbruster1, Neil Wilson2, Ravi Prakash Reddy Nanga1, and Ravinder Reddy1
1Radiology, University of Pennsylvania, Philadelphia, PA, United States, 2Siemens Medical Solutions USA Inc, Malvern, PA, United States
Intracellular acidosis, mainly due to lactic acid accumulation, has been regarded as the major contributor of skeletal muscle fatigue and may be a contributing factor across many musculoskeletal disorders, such as peripheral arterial disease (PAD), Duchenne dystrophy and Becker dystrophy, primary mitochondrial disorders, diabetes mellitus, and cardiovascular disease. Hence, an imaging biomarker capable of inferring the underlying acidification during varying exercise conditions could be valuable in assessing the efficacy of potential therapy options. Here, we demonstrate the feasibility of indirect detection of acidification in exercised skeletal muscle using creatine CEST.
|
|||
1459.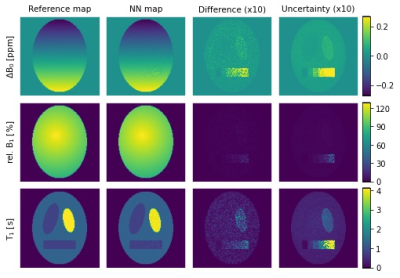 |
Simultaneous mapping of B0, B1 and T1 for the correction of CEST-MRI
Kerstin Heinecke1, Henrik Narvaez1, Christoph Kolbitsch1, and Patrick Schuenke1
1Physikalisch-Technische Bundesanstalt (PTB), Braunschweig and Berlin, Germany
One of the bottlenecks in Chemical Exchange Saturation Transfer (CEST) MRI is its susceptibility to magnetic field inhomogeneities and dependency on T1 relaxation times. To enable efficient correction for these parameters, we propose a new method for simultaneous quantitative mapping of B0, B1 and T1 using a simple CEST sequence with a modified preparation block. For the analysis, we developed a neural network that provides an additional uncertainty estimation for each parameter. First results on phantom simulations demonstrate the feasibility of this approach as well as its applicability for the correction of CEST-MRI data.
|
|||
1460.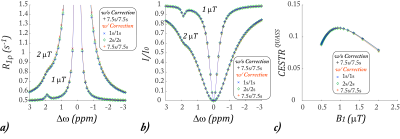 |
Quasi-steady-state (QUASS) CEST solution improves the accuracy of CEST quantification – QUASS CEST MRI-based omega plot analysis
Phillip Zhe Sun1
1Department of Radiology and Imaging Sciences, Emory University, Atlanta, GA, United States
CEST MRI omega plot quantifies the labile proton fraction ratio (fr) and exchange rate (ksw), yet it assumes long RF saturation time (Ts) and relaxation delay (Td). Our study aimed to test if a quasi-steady-state (QUASS) CEST analysis that accounts for the effect of finite Ts and Td could improve the accuracy of CEST MRI quantification. Both simulation and CEST MRI experiments confirmed that the QUASS solution enabled robust quantification of ksw and fr, superior over the omega plot analysis based on the conventional apparent CEST MRI measurements.
|
|||
1461.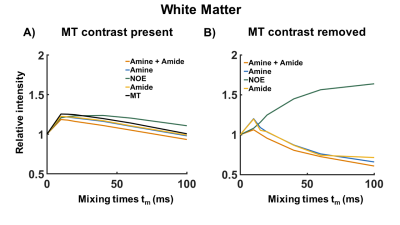 |
MTC removed and exchange rate differentiated CEST using Variable Delay Multi Pulse (VDMP) in the human brain at 7T
Bárbara Schmitz Abecassis1, Elena Vinogradov Vinogradov2,3, Jannie P. Wijnen4, Thijs van Harten1, Evita C. Wiegers4, Hans J.M. Hoogduin4, Matthias J.P. van Osch 1, and Ece Ercan1
1Department of Radiology, Leiden University Medical Center, Leiden, Netherlands, 2Department of Radiology, UT Southwestern Medical Center, Dallas, TX, United States, 3Advanced Imaging Research Center, UT Southwestern Medical Center, Dallas, TX, United States, 4Department of Radiology, University Medical Center Utrecht, Utrecht, Netherlands
Variable-delay multi-pulse (VDMP) is a chemical exchange saturation transfer (CEST) editing technique relying on the exchange rate as a filter to separate the CEST- effect from different solute pools. We investigated this method to evaluate CEST-effects from metabolites and proteins in in vivo human brain at 7T. We removed the pronounced magnetization transfer contrast present in vivo via Lorentzian-fittings, and evaluated the effect of B1 on saturation build-up curves of the different CEST-pools. MTC-removed VDMP-curves from the human brain are reported for the first time, where the effect of slow and fast exchanging protons allows distinguishing different CEST-pools.
|
|||
1462.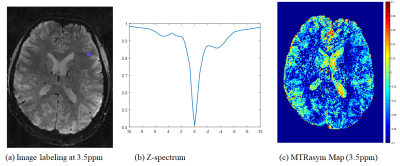 |
Highly Accelerated 1mm3-Isotropic 3D CEST MRI with Spectral Random Walk CAIPIRINHA Sampling at 7T
Sugil Kim1, Seong-Gi Kim2,3, and Suhyung Park4,5
1Siemens Healthineers, Seoul, Korea, Republic of, 2Center for Neuroscience Imaging Research (CNIR), Institute for Basic Science (IBS), Suwon, Korea, Republic of, 3Department of Biomedical Engineering, Sungkyunkwan University, Suwon, Korea, Republic of, 4Department of Computer Engineering, Chonnam National University, Gwangju, Korea, Republic of, 5Department of ICT Convergence System Engineering, Chonnam National University, Gwangju, Korea, Republic of
We propose highly accelerated 3D CEST MRI using CAIPI sampling based 3D segmented EPI with spectral random walk, potentially enabling 1mm-isotropic whole-brain CEST imaging within 5min at 7T.
|
|||
1463.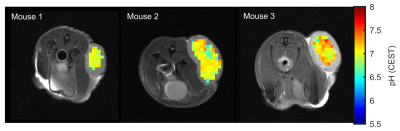 |
Mapping of intracellular pH in vivo using amide and guanidyl CEST-MRI at 9.4 T
Philip S Boyd1, Johannes Breitling1, Stephanie Laier2, Karin Mueller-Decker2, Andrey Glinka3, Mark E Ladd1, Peter Bachert1, and Steffen Goerke1
1Division of Medical Physics in Radiology, German Cancer Research Center (DKFZ), Heidelberg, Germany, 2Center for Preclinial Research, Core Facility Tumor Models, German Cancer Research Center (DKFZ), Heidelberg, Germany, 3Division of Molecular Embryology, German Cancer Research Center (DKFZ), Heidelberg, Germany
In this study, we present a method for mapping intracellular pH values based on amide and guanidyl CEST-MRI which simultaneously compensates for concentration changes, the semi-solid magnetization transfer, and spillover dilution. Compensation of the concomitant effects and calculation of reliable pH maps in the range of pH 6.5-7.5 was verified experimentally in porcine brain lysates. Applicability in vivo was demonstrated in the lesions of three tumor-bearing mice showing an average intracellular pH of approximately 7. Consequently, endogenous amide and guanidyl signal-based mapping of the intracellular pH is now applicable for tumor imaging which was previously prevented by the concomitant effects.
|
|||
1464.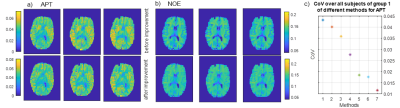 |
7 tricks for 7T CEST: improving the reproducibility of multi-pool evaluation
Angelika Mennecke1, Katrin Khakzar1, Kai Herz2, Moritz Fabian1, Alexander German1, Andrzej Liebert3, Ingmar Blümcke4, Burkhard Kasper5, Manuel Schmidt1, Arnd Dörfler1, Armin Nagel3, Frederik Laun3, and Moritz Zaiß1
1Department of Neuroradiology, University Hospital of Erlangen, FAU Erlangen-Nürnberg, Erlangen, Germany, 2High-field Magnetic Resonance Center, Max Planck Institute for Biological Cybernetics, Tübingen, Germany, 3Institute of Radiology, University Hospital of Erlangen, FAU Erlangen-Nürnberg, Erlangen, Germany, 4Institute of Neuropathology, University Hospital of Erlangen, FAU Erlangen-Nürnberg, Erlangen, Germany, 5Department of Neurology, Epilepsy Centre, University Hospital of Erlangen, FAU Erlangen-Nürnberg, Erlangen, Germany
With the help of the presented post-processing procedure, it is possible to obtain an increased reproducibility. For the amide contrast, the CoV decreased from 4% to less than 1.2 %.
|
|||
1465.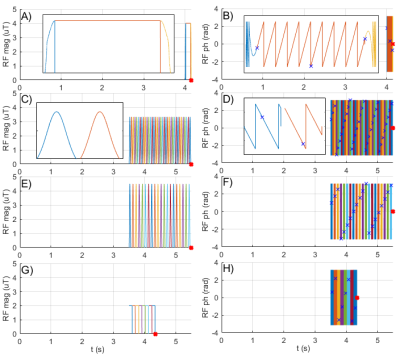 |
Pulseq-CEST: Towards multi-site multi-vendor compatibility and reproducibility of CEST experiments using an open source sequence standard
Kai Herz1,2, Sebastian Mueller1, Maxim Zaitsev3, Linda Knutsson4,5, Jinyuan Zhou5, Phillip Zhe Sun6, Peter van Zijl5,7, Klaus Scheffler1,2, and Moritz Zaiss1,8
1Magnetic Resonance Center, MPI for Biological Cybernetics, Tuebingen, Germany, 2Biomedical Magnetic Resonance, University of Tuebingen, Tuebingen, Germany, 3Center for Medical Physics and Biomedical Engineering, Medical University of Vienna, Vienna, Austria, 4Medical Radiation Physics, Lund University, Lund, Sweden, 5Russell H. Morgan Department of Radiology and Radiological Science, Johns Hopkins University School of Medicine, Baltimore, MD, United States, 6Yerkes Imaging Center, Emory University, Atlanta, GA, United States, 7F.M. Kirby Research Center for Functional Brain Imaging, Kennedy Krieger Institute, Baltimore, MD, United States, 8Neuroradiology, Friedrich‐Alexander Universität Erlangen‐Nuernberg, Erlangen, Germany The design of the preparation period is crucial for a CEST experiment, thus, a common, easy-to-use and vendor-independent file format to share exact preparation parameters is desirable. Here, we propose the use of Pulseq to define CEST parameters in an open source and human-readable file format. By providing a protocol database, a pulseq-compatible Bloch-McConnell simulation and a hybrid sequence using Pulseq for CEST preparation, we present a straightforward approach to standardize, share, simulate and measure different CEST preparation schemes, which are inherently completely defined. |
|||
1466.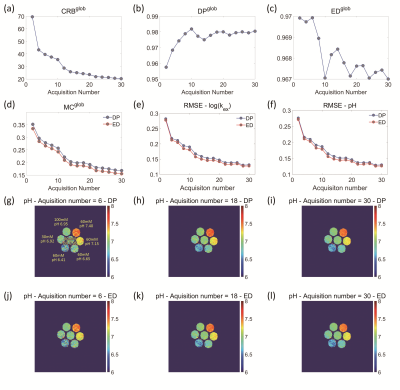 |
Assessment of acquisition strategies of CEST MR fingerprinting pH imaging using Cramer-Rao Bound algorithm
Jie Liu1, Hui Liu2, Qi Liu2, Jian Xu2, Hairong Zheng1, and Yin Wu1
1Paul C. Lauterbur Research Center for Biomedical Imaging, Shenzhen Institutes of Advanced Technology, Chinese Academy of Sciences, Shenzhen, China, 2United Imaging Healthcare America, Houston, TX, United States
Optimization of acquisition strategies plays a key role in CEST-MRF imaging. However, an effective method to assess the performance of acquisition strategies in CEST quantification is lacking. In this study, we proposed a Cramer-Rao bound (CRB) based metric to measure the accuracy of CEST-MRF in pH quantification under various acquisition schemes. The results of both simulation and phantom experiments showed that the CRB-based index outperformed the conventionally used dictionary similarity metrics that are measured with dot products and Euclidean distance, implying that the proposed algorithm has great potential in optimizing CEST-MRF imaging for improved pH measurement.
|
|||
1467.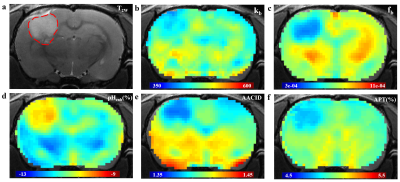 |
A comparison of several tumor endogenous pH mapping methods using CEST at 11.7T
Ying Liu1,2, Botao Zhao1,2, and Xiao-Yong Zhang1,2
1Institute of Science and Technology for Brain-Inspired Intelligence, Fudan University, Shanghai, China, 2Key Laboratory of Computational Neuroscience and Brain-Inspired Intelligence (Fudan University), Ministry of Education, Shanghai, China
CEST-MRI has emerged as a noninvasive pH imaging technique based on the pH-dependence of the prototropic exchange rates. In this work, we compared several recently published CEST pH imaging methods and proton exchange rate value in rat glioma models at 11.7T. All of these pH-weighted contrasts show a pH correlation and have significantly different signal intensities between the tumor region and the surrounding normal tissue. Our results demonstrated that pH enhanced method and APT imaging are not concentration-independent while the sensitivity of AACID is affected by the weak amine signals at 2.7ppm.
|
|||
1468.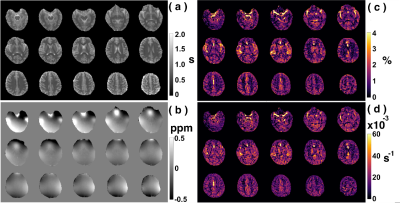 |
Whole-brain amide CEST at 3T with a steady-state radial MRI acquisition
Ran Sui1,2,3, Lin Chen1,2, Yuguo Li1,2, Jianpan Huang4, Kannie W.Y Chan2,4, Xiang Xu5, Peter C.M.van Zijl1,2, and Jiadi Xu1,2
1F.M. Kirby Research Center for Functional Brain Imagin, Kennedy Krieger Institute, Baltimore, MD, United States, 2Russell H. Morgan Department of Radiology and Radiological Science, Johns Hopkins University School of Medicine, Baltimore, MD, United States, 3Department of Biomedical Engineering, Johns Hopkins University, Baltimore, MD, United States, 4Department of Biomedical Engineering, City University of Hong Kong, Hong Kong, China, 5BioMedical Engineering and Imaging Institute, Icahn School of Medicine at Mount Sinai, New York, NY, United States
We aim to develop an acquisition and processing pipeline for amideCEST mapping on human brain at 3T. This includes steady-state radial CEST (starCEST) using PROPELLOR-based sampling to acquire Z-spectra that are robust to brain motions and one multi-linear singular value decomposition (MLSVD) post-processing for enhancing the CEST SNR. Finally, the amideCEST peak was quantified using the polynomial and Lorentzian line-shape fitting (PLOF) approach.The amideCEST contrast extracted by the PLOF method trended towards a stronger gray matter (GM) signal (1.33% ± 0.34%) than white matter (WM) (0.92% ± 0.09%; p=0.06, n=4).
|
|||
1469.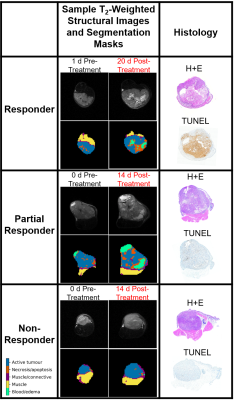 |
Investigating MT and CEST Characteristics of DU145 Prostate Tumour Xenografts in Relation to Radiation Treatment Response
Leedan Murray1, Wilfred W. Lam1, and Greg J. Stanisz1,2
1Physical Sciences, Sunnybrook Research Institute, Toronto, ON, Canada, 2University of Toronto, Toronto, ON, Canada
MT and CEST characteristics of DU145 prostate tumour xenografts with known responses to radiation treatment were investigated to determine characteristics that may predict treatment response. After excision for histology, tumours were categorized according to their response to treatment (responder, partial responder and non-responder). Average MTR, qMT parameters, and CEST and rNOE contributions were calculated for each category. Significant changes were mostly observed in the necrotic/apoptotic regions of the tumour and were most evident in the qMT parameters. It may be possible to determine efficacy early on in the course of treatment based on the studied MR characteristics.
|
|||
1470.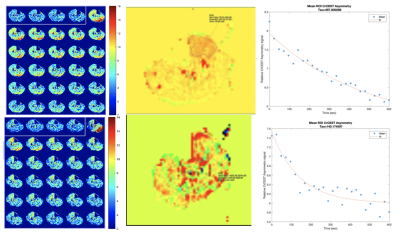 |
An ASL and CrCEST combined protocol at 3T in the Study of Metabolic and Perfusion Changes Post Revascularization in Peripheral Arterial Disease
Helen Sporkin1, Toral Patel2, Christopher Schumann2, Christopher Kramer2,3, and Craig Meyer1,3
1Biomedical Engineering, University of Virginia, Charlottesville, VA, United States, 2Cardiovascular Medicine, University of Virginia, Charlottesville, VA, United States, 3Radiology and Medical Imaging, University of Virginia, Charlottesville, VA, United States
Patients with peripheral arterial disease (PAD) have been shown to have a delayed phosphocreatine recovery due to chronic ischemia. Creatine chemical exchange saturation transfer (CrCEST) is an MRI technique that allows for creatine concentrations to be monitored at high spatial resolution. Combining ASL and CrCEST in the same protocol allows for spatial visualization of metabolism and perfusion. The goal of this study is to use this combination to assess changes after revascularization in patients undergoing both endovascular and surgical procedures and evaluate the recovery of both perfusion and metabolism in their muscle tissue.
|
The International Society for Magnetic Resonance in Medicine is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.