Digital Posters
Relaxometry in the Body: MSK & More
ISMRM & SMRT Annual Meeting • 15-20 May 2021

| Concurrent 1 | 19:00 - 20:00 |
3305.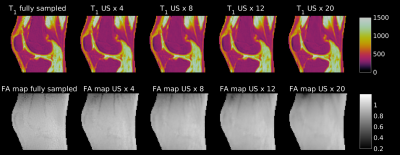 |
Accelerated 3D-UTE-AFI B1 mapping to correct VFA-based T1 estimations in short T2* tissues
Marta Brigid Maggioni1, Martin Krämer1,2, and Jürgen R. Reichenbach1,2,3,4,5
1Medical Physics Group, Institute of Diagnostic and Interventional Radiology, Jena University Hospital - Friedrich Schiller University, Jena, Germany, 2Institute of Diagnostic and Interventional Radiology, Jena University Hospital - Friedrich Schiller University, Jena, Germany, 3Michael Stifel Center for Data-driven and Simulation Science Jena, Friedrich Schiller University, Jena, Germany, 4Abbe School of Photonics, Friedrich Schiller University, Jena, Germany, 5Center of Medical Optics and Photonics, Friedrich Schiller University, Jena, Germany
B1 mapping is a considerable challenge for T1 quantification, and it is especially difficult for short T2* species because only ultrashort echo-time (UTE) imaging sequences are able to retrieve signal in such tissues. Recently a method called Actual Flip Angle Imaging (AFI) has shown promising results because of its potential to be combined with UTE acquisition. However the AFI method is characterized by long acquisition times. In this work we propose to use undersampling to reduce the acquisition times of AFI-based B1 mapping.
|
|||
3306.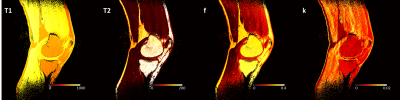 |
Feasibility of high resolution quantitative magnetic resonance imaging using variable flip angle and spoiling phase angle
Refaat E Gabr1, Lingzhi Hu2, Xingxian Shou2, Yongquan Ye2, Weiguo Zhang2, and Ponnada A Narayana1
1Diagnostic and Interventional Imaging, University of Texas Health Science Center at Houston, Houston, TX, United States, 2UIH America Inc., Houston, TX, United States
We developed a multi-flip multi-spoiling phase angle method for quantitative assessment of tissue parameters. Application in phantom studies and in vivo knee imaging shows promising performance for high resolution parameter mapping in clinically feasible scan time.
|
|||
3307.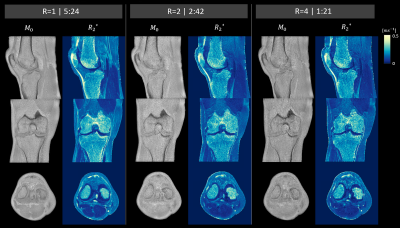 |
CUTE: Compressed Sensing UTE for Multi-Echo T2* Mapping
Stefan Sommer1,2, Tom Hilbert3,4,5, Constantin von Deuster1,2, Natalie Hinterholzer2, Markus Klarhöfer1, and Daniel Nanz2,6
1Siemens Healthcare, Zurich, Switzerland, 2Swiss Center for Musculoskeletal Imaging (SCMI), Balgrist Campus, Zurich, Switzerland, 3Advanced Clinical Imaging Technology (ACIT), Siemens Healthcare, Lausanne, Switzerland, 4Department of Radiology, Lausanne University Hospital (CHUV), Lausanne, Switzerland, 5LTS5, École Polytechnique Fédérale de Lausanne, Lausanne, Switzerland, 6University of Zurich, Zurich, Switzerland
Clinical application of UTE (ultrashort echo time) sequences in musculoskeletal studies remains limited, despite their potential to unveil quantitative short-T2* information of e.g. tendons or ligaments. The major shortcoming of quantitative mapping is the considerably longer scan time compared to conventional, anatomical imaging sequences. We investigated the feasibility and use of an isotropic 3D radial compressed sensing UTE (CUTE) prototype sequence for quantitative mapping of short-T2* components in the knee. Similar image quality and T2* values were obtained for non-accelerated and accelerated acquisitions. Thus, CUTE sequences show great promise for fast and clinically feasible UTE T2* mapping.
|
|||
 |
3308.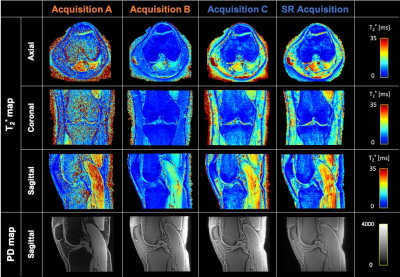 |
Super-resolution T2* mapping of the knee using UTE Spiral VIBE MRI
Céline Smekens1, Quinten Beirinckx2, Floris Vanhevel3, Pieter Van Dyck3, Arjan den Dekker2, Jan Sijbers2, Thomas Janssens1, and Ben Jeurissen2
1Siemens Healthcare NV/SA, Beersel, Belgium, 2imec-Vision Lab, Department of Physics, University of Antwerp, Wilrijk, Belgium, 3Department of Radiology, Antwerp University Hospital and University of Antwerp, Edegem, Belgium
T2* mapping using ultrashort echo time (UTE) MRI allows for quantitative evaluation of collagen-rich knee structures with short mean transverse relaxation times. However, acquisitions with low through-plane resolution are commonly used to obtain T2* maps within reasonable scan times, affecting the accuracy of the estimations because of partial volume effects. In this work, model-based super-resolution reconstructions based on UTE Spiral VIBE MRI were performed to obtain high-resolution T2* maps of knee structures within a reasonable scan time. The obtained T2* maps are comparable to maps generated with direct 3D UTE Spiral VIBE acquisitions while requiring approximately 25% less scan time.
|
||
3309.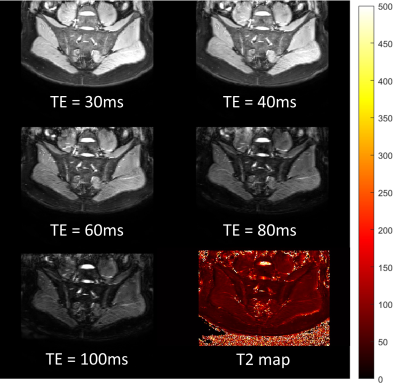 |
Fat-insensitive T2water measurement using multiple Dixon turbo spin-echo acquisitions with effective echo time increments
Ruaridh M Gollifer1,2, Tim JP Bray1,3, Margaret Hall-Craggs1,3, and Alan Bainbridge2
1Centre for Medical Imaging (CMI), University College London, London, United Kingdom, 2Department of Medical Physics and Bioengineering, University College London Hospital, London, United Kingdom, 3Department of Imaging, University College London Hospital, London, United Kingdom
Short tau inversion recovery (STIR) imaging is the mainstay of clinical imaging in inflammatory musculoskeletal (MSK) diseases, and generates hyperintense signal in areas of inflammation in bone marrow (bone marrow oedema). However, the assessment of STIR images of bone marrow is based on qualitative judgement of signal intensity and can be confounded by variations in fat content resulting from the healing response in bone to inflammation. We aimed to develop a method which would (1) separate fat and water and (2) provide a water-specific T2 measurement, enabling separation and individual quantification of oedema and fat in the bone marrow.
|
|||
3310.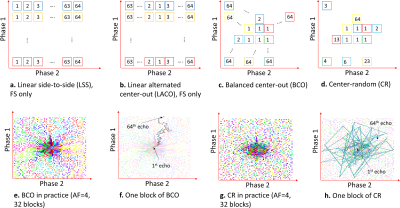 |
Prospective Accelerated Cartesian 3D-T1rho Mapping of Knee Joint using Data-Driven Optimized Sampling Patterns and Compressed Sensing
Marcelo Victor Wust Zibetti1, Azadeh Sharafi1, Mahesh Bharath Keerthivasan2, and Ravinder Regatte1
1Radiology, NYU Langone Health, New York, NY, United States, 2Siemens Healthineers, New York, NY, United States
We modified the Cartesian 3D-fast spoiled gradient-echo sequence with T1rho magnetization preparation for prospective acceleration of knee-joint mapping using optimized sampling patterns (SPs) and compressed sensing (CS) reconstructions. In this sequence, after each T1rho preparation module, several k-space lines are captured, partially filling the 3D k-space. However, the ordering of the k-space filling is very important to maintain consistent T1rho contrast and to obtain stable quantitative mapping. This is even more challenging when arbitrary SPs are used for accelerated MRI. We investigate different k-space ordering schemes considering optimized SPs and Poisson disk SPs in prospective 3D-T1rho acquisition with CS reconstructions.
|
|||
3311.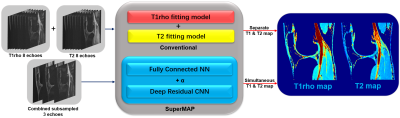 |
SuperMAP: Superfast MR Mapping with Joint Under-sampling using Deep Combined Network
Hongyu Li1, Mingrui Yang2, Jeehun Kim2, Chaoyi Zhang1, Ruiying Liu1, Peizhou Huang3, Sunil Kumar Gaire1, Dong Liang4, Xiaoliang Zhang3, Xiaojuan Li2, and Leslie Ying1,3
1Electrical Engineering, University at Buffalo, State University of New York, Buffalo, NY, United States, 2Program of Advanced Musculoskeletal Imaging (PAMI), Cleveland Clinic, Cleveland, OH, United States, 3Biomedical Engineering, University at Buffalo, State University of New York, Buffalo, NY, United States, 4Paul C. Lauterbur Research Center for Biomedical Imaging, Medical AI research center, SIAT, CAS, Shenzhen, China
This abstract presents a combined deep learning framework SuperMAP to generate MR parameter maps from very few subsampled echo images. The method combines deep residual convolutional neural networks (DRCNN) and fully connected networks (FC) to exploit the nonlinear relationship between and within the combined subsampled T1rho/T2 weighted images and the combined T1rho/T2 maps. Experimental results show that the proposed combined network is superior to single CNN network and can generate accurate T1rho and T2 maps simultaneously from only three subsampled echoes within one scan with results comparable to reference from fully sampled 8-echo images each for two separate scans.
|
|||
3312.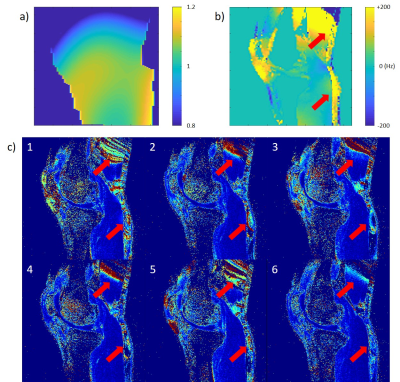 |
MR T1ρ preparations: B1 and B0 inhomogeneity response on 3T and 7T systems
Jeehun Kim1,2, Qi Peng3, Can Wu4,5, and Xiaojuan Li1,6
1Department of Biomedical Engineering, Program of Advanced Musculoskeletal Imaging (PAMI), Cleveland Clinic, Cleveland, OH, United States, 2Case Western Reserve University, Cleveland, OH, United States, 3Department of Radiology, Albert Einstein College of Medicine and Montefiore Medical Center, Bronx, NY, United States, 4Department of Medical Physics, Memorial Sloan Kettering Cancer Center, New York, NY, United States, 5Philips Healthcare, Andover, MA, United States, 6Department of Diagnostic Radiology, Cleveland Clinic, Cleveland, OH, United States
Quantitative T1ρ mapping is a promising biomarker for detecting tissue compositional changes at early stages of diseases. For reliable and reproducible measurements, T1ρ preparation pulses should be robust to B0 and B1 inhomogeneity. In this work, six different preparation schemes were evaluated in terms of their responses to B0 and B1 inhomogeneities on agarose phantoms and volunteers using both 3T and 7T MRI systems.
|
|||
3313.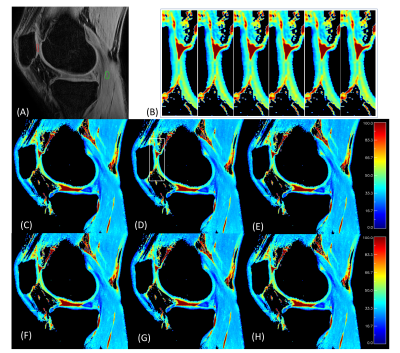 |
Efficient Phase Cycling Strategy for High Resolution Three-Dimensional GRE Quantitative Mapping
Qi Peng1, Can Wu2,3, Jee Hun Kim4,5, and Xiaojuan Li4,5,6
1Department of Radiology, Albert Einstein College of Medicine, Bronx, NY, United States, 2Department of Medical Physics, Memorial Sloan Kettering Cancer Center, New York, NY, United States, 3Philips Healthcare, Andover, MA, United States, 4Program of Advanced Musculoskeletal Imaging (PAMI), Cleveland Clinic, Cleveland, OH, United States, 5Department of Biomedical Engineering, Cleveland Clinic, Cleveland, OH, United States, 6Department of Diagnostic Radiology, Cleveland Clinic, Cleveland, OH, United States
Magnetization-prepared gradient echo (MP-GRE) sequences have been commonly used for quantitative MRI in the literature to improve imaging speed. Paired acquisitions with RF phase-cycling could eliminate image blurring due to longitudinal relaxation and loss of MP contrast along the GRE readouts, with doubled scan time. This study introduces a novel unpaired phase cycling strategy to eliminate the time penalty with reduced sensitivity to B0 field inhomogeneities for high resolution quantitative mapping in MP-GRE sequences. The feasibility and efficacy of this strategy were demonstrated in both phantom and human studies.
|
|||
3314.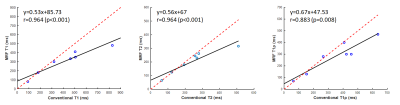 |
The feasibility of T1ρ magnetic resonance fingerprinting with a random design of T1ρ preparation at 11.7T
Qianfeng Wang1, He Wang1,2, Danyang Feng1, Fei Dai1, Yuwen Zhang1, and Baofeng Yang1
1Institute of Science and Technology for Brain-Inspired Intelligence, Fudan University, Shanghai, China, 2Human Phenome Institute, Fudan University, Shanghai, China, Shanghai, China
In this study, we demonstrate that T1ρ-MRF can be achieved by varying flip angle, repetition time, echo time, as well as spin lock time—in a pseudorandom manner at 11.7T MR system.
|
|||
3315.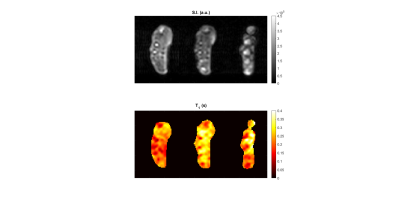 |
In vivo $$$T_1$$$ quantification at 0.1 T using a fast, interleaved Look-Locker based $$$T_1$$$ mapping sequence.
Marco Fiorito1, Maksym Yushchenko1, Davide Cicolari2, Mathieu Sarracanie1, and Najat Salameh1
1Center for Adapatable MRI Technology (AMT center), Department of Biomedical Engineering, University of Basel, Allschwil, Switzerland, 2Department of Physics, University of Pavia, Pavia, Italy
$$$T_1$$$ mapping in MRI can be employed in a variety of techniques for diagnosis and treatment follow-up, but generally suffers from long acquisition times. Little effort has been put in low magnetic field regimes, where sensitivity is further impeded. Nevertheless, low field provides higher $$$T_1$$$ dispersion and favours adaptable scanner designs, suitable for dedicated applications such as MRI of body extremities. Here, we assess our newly developed interleaved, Look-Locker based $$$T_1$$$ mapping sequence in calibrated samples, and we present a first in vivo $$$T_1$$$ map of a volunteer’s hand at 0.1 T.
|
|||
3316.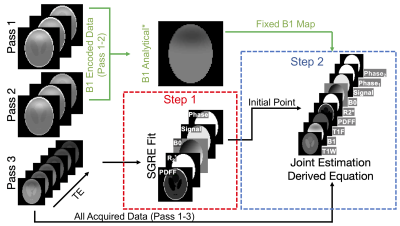 |
Simultaneous Fat- and B1-Corrected T1 Mapping Using Chemical-Shift Encoded MRI
Nathan Tibbitts Roberts1,2, Diego Hernando1,3, Daiki Tamada1, and Scott B Reeder1,3,4,5,6
1Radiology, University of Wisconsin - Madison, Madison, WI, United States, 2Electrical and Computer Engineering, University of Wisconsin - Madison, Madison, WI, United States, 3Medical Physics, University of Wisconsin - Madison, Madison, WI, United States, 4Biomedical Engineering, University of Wisconsin - Madison, Madison, WI, United States, 5Medicine, University of Wisconsin - Madison, Madison, WI, United States, 6Emergency Medicine, University of Wisconsin - Madison, Madison, WI, United States
Tissue fat and spatially varying B1 inhomogeneities are known confounders of quantitative T1W(ater) mapping methods that use variable flip angle techniques. T1W measurements can be corrected for B1 heterogeneity, but this typically requires an additional B1 calibration acquisition both extending patient acquisition time and introducing image registration considerations. In this work we propose simultaneous estimation of B1, T1W, proton density fat-fraction and R2* using a three-pass approach using dual orthogonal RF pulses and multiple flip angles. The feasibility and noise performance of this proposed acquisition and fitting strategy are evaluated using Cramer-Rao Lower Bound analysis, simulations, and phantom experiments.
|
|||
3317.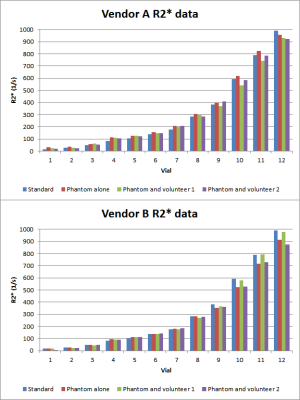 |
Inter-vendor 3T R2* mapping evaluation on a standardized R2* phantom with and without a human subject
Justin Yu1, Anshuman Panda1, and Alvin Silva1
1Department of Radiology, Mayo Clinic Arizona, Phoenix, AZ, United States
Quantitative MRI can precisely measure biomarkers for disease, such as liver hemochromatosis. R2* correlates with liver iron concentration (LIC), but data is scarce for 3T R2* mapping. The out-of-the box vendor provided sequences for quantitative R2* measurements do not precisely measure large R2* values in a standardized phantom. The discrepancy is greater at higher R2* values. This can lead to underestimates in LIC quantification for patients with severe hemochromatosis. R2* mapping from different MRI vendors may yield different results, and patient specific QA may be necessary for clinical utilization for quantitative R2* mapping.
|
|||
3318.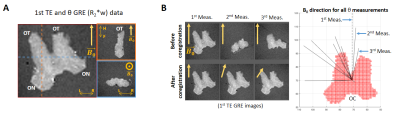 |
Effects of fibre dispersion and myelin content on R2*: simulations and post-mortem experiments
Francisco Javier Fritz1, Mohammad Ashtarayeh1, Joao Periquito2, Andreas Pohlmann2, Markus Morawski3, Carsten Jaeger4, Thoralf Niendorf2, Kerrin J. Pine4, Evgeniya Kirilina4,5, Nikolaus Weiskopf4,6, and Siawoosh Mohammadi1
1Institut für Systemische Neurowissenschaften, Universitätklinikum Hamburg-Eppendorf, Hamburg, Germany, 2Berlin Ultrahigh Field Facility (B.U.F.F.), Max-Delbrueck-Center for Molecular Medicine in the Helmholtz Association, Berlin, Germany, 3Paul Flechsig Institute of Brain Research, University of Leipzig, Leipzig, Germany, 4Department of Neurophysics, Max Planck Institute for Human Cognitive and Brain Sciences, Leipzig, Germany, 5Center for Cognitive Neuroscience Berlin, Free University Berlin, Berlin, Germany, 65Felix Bloch Institute for Solid State Physics, Faculty of Physics and Earth Sciences, Leipzig University, Leipzig, Germany
We studied the impact of fibre dispersion and myelin on the angle-dependent gradient-recalled echo signal decay in simulation and experimental data from post-mortem tissue. We compared the classical log-mono-exponential and quadratic time-dependent signal model (M2) derived from Wharton and Bowtell’s forward-model with and without myelin-water contribution. We found that R2*-angular dependency was modulated by fibre dispersion and the R2*-angular dependency is removed using M2. We also observed that the higher-order parameter estimated from experimental data at small angles and dispersion was only reflected in simulations when accounting for myelin-water contributions, indicating that this pool needs to be added into M2.
|
|||
3319.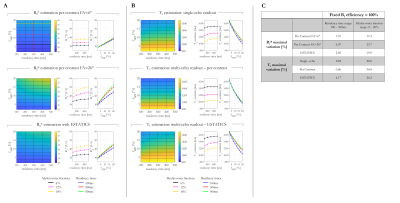 |
The impact of multi-compartment microstructure on single-compartment T1 estimates
Giorgia Milotta1, Nadège Corbin1,2, Antoine Lutti3, Siawoosh Mohammadi4,5, and Martina Callaghan1
1Wellcome Centre for Human Neuroimaging, UCL Queen Square Institute of Neurology, University College London, London, United Kingdom, 2Centre de Résonance Magnétique des Systèmes Biologiques, UMR5536, CNRS/University Bordeaux, Bordeaux, France, 3Laboratory for Research in Neuroimaging, Department for Clinical Neuroscience, Lausanne University Hospital and University of Lausanne, Lausanne, Switzerland, 4Department of Systems Neurosciences, University Medical Center Hamburg-Eppendorf, Hamburg, Germany, 5Department of Neurophysics, Max Planck Institute for Human Cognitive and Brain Sciences, Leipzig, Germany
Quantitative relaxometry in the brain is appealing because of its microstructural sensitivity. Estimating bulk parameters assumes a single relaxation time per voxel, which is only valid when the residency time is short with respect to T1. In reality, the relative contribution of sub-compartments will depend on flip angle and echo time. Here, we simulate a two-compartment model (myelin and intra-extracellular water) and estimate T1 with FA-specific signals derived via three estimation schemes. We quantify the impact of myelin water fraction, residency time and transmit field inhomogeneity on these estimates and find good correspondence with in vivo T1 estimates at 7T.
|
|||
3320.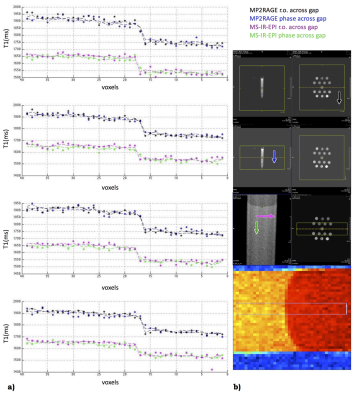 |
Towards in-vivo myeloarchitecture: optimising T1 maps point spread function by very high resolution multi-shot inversion-recovery EPI
Fabrizio Fasano1,2, John Evans3, Chloe Benson4, Yifei Wang4, Derek K Jones3,5, Alison Paul4, and Robert Turner6,7
1Siemens Healthcare Ltd, Camberly, United Kingdom, 2Siemens Healthcare GmbH, Erlangen, Germany, 3Cardiff University Brain Research Imaging Centre (CUBRIC), Cardiff University, Cardiff, United Kingdom, 4School of Chemistry, Cardiff University, Cardiff, United Kingdom, 5Mary McKillop Institute for Health Research, Faculty of Health Sciences, Australian Catholic University, Melbourne, Australia, 6Department of Neurophysics, Max Planck Institute for Human Cognitive and Brain Sciences, Leipzig, Germany, 7Sir Peter Mansfield Imaging Centre, University of Nottingham, Nottingham, United Kingdom
Myeloarchitecture has a critical role in the specialisation of brain microcircuitry. It shapes network functions from small to large scale, and ultimately behaviour. We tested, on homemade Agar gel phantoms, the multi-shot inversion recovery slice shuffled EPI IR-MS-EPI approach recently proposed by Sanchez and co-workers (Proc. Intl. Soc. Mag. Reson. Med. 2018, 60). Our measurement results and the simulated performances of MS-IR-EPI showed a good reproducibility of the T1 maps and a preserved quality of its point spread function, when acquiring at higher-than 500μm in-plane resolution, making it suitable for assessing grey matter myelination process.
|
|||
3321.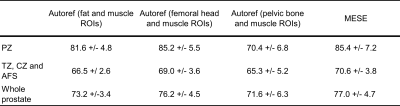 |
Pseudo-T2 mapping of T2-weighted MRI of the prostate: Comparison to gold standard
Kaia Ingerdatter Sørland1, Pål Erik Goa2,3, Kirsten Margrete Selnæs1,3, Elise Sandsmark3, Cristopher George Trimble1, Mohammed R. S. Sunoqrot1, Mattijs Elschot1,3, and Tone F. Bathen1,3
1Department of Circulation and Medical Imaging, Norwegian University of Science and Technology, Trondheim, Norway, 2Department of Physics, Norwegian University of Science and Technology, Trondheim, Norway, 3Department of Radiology and Nuclear Medicine, St. Olavs University Hospital, Trondheim, Norway
Quantitative analysis of T2-weighted (T2W) MR images is hindered by the lack of signal intensity standardization. The tissue T2 values are independent of variations in scanner parameters, but T2 mapping is normally not part of the clinical pathway. Autoref is an automated dual-reference tissue normalization of T2W images developed in our group, aiming to reproduce the prostate T2 values. In this study we measured the prostate T2 value in seven healthy volunteers, and compared them with pseudo-T2 values from Autoref normalized T2W. The Autoref was proven to reproduce prostate T2 values as well as contrast within the prostate zones.
|
|||
3322.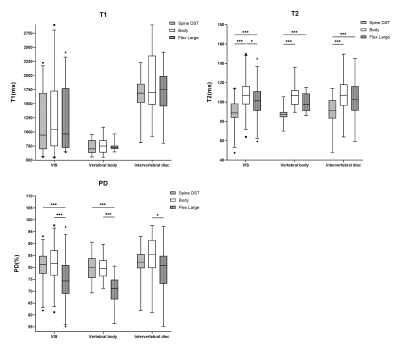 |
Reliability and reproducibility of synthetic spine MRI with different coils
Yitong Li1, Xiaoqing Liang1, Bowen Hou1, Yan Xiong1, Weiyin Vivian Liu2, and Xiaoming Li1
1Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China, 2MR Research, GE Healthcare, Beijing, China
Multiple relaxation quantitative maps can be obtained from a single scan with synthetic MRI. Previous researches have investigated this technique in lumbar spine applications, but coil selection, which may introduce bias into the quantitative data, has not been taken into account. In this study, we compared the measured quantitative parameters (T1, T2, and PD) using three coils separately and evaluated data reliability and repeatability. Coil selection contributes to differences in measurements of lumbar spine synthetic MRI, which should be concerned in future studies.
|
|||
3323.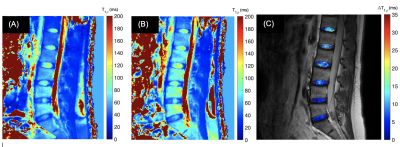 |
T1rho Dispersion Imaging of Intervertebral Discs
Ping Wang1, Jay D Turner1, Juan Uribe1, and John C Gore2
1Barrow Neurological Institute, Phoenix, AZ, United States, 2Institute of Imaging Science, Vanderbilt University, Nashville, TN, United States
This study aims to develop a T1ρ dispersion protocol method for human lumbar spine imaging, with a specific goal to detect proteoglycan loss in the intervertebral disc (IVD), which is an initial step in degenerative disc disease (DDD). T1ρ dispersion parametric images are able to distinguish the nucleus pulposus from annulus fibrosus based on differences in exchangeable protons, as predicted theoretically. This work is relevant for clinical applications for DDDas it provides novel, quantitative, and clinically translatable in vivo imaging biomarkers capable of detecting proteoglycan changes.
|
|||
3324.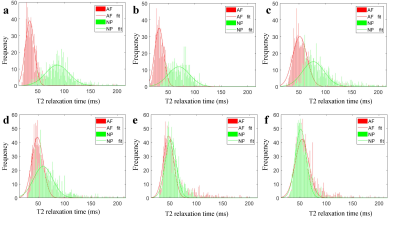 |
Distinction of T2 quantitative measurements between the nucleus pulposus and anulus fibrosus using Gaussian-fitted histogram analysis
Xiaoqing Liang1, Weiyin Vivian Liu2, Jingyi Wang1, and Xiaoming Li1
1Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China, 2MR Research,GE Healthcare, Beijing, China
The distinction between the nucleus pulposus (NP) and the peripheral annulus fibrosus (AF) of intervertebral disc is an important diagnostic indicators of intervertebral disc degeneration (IVDD) on MRI. So far, there have not been many studies or mature approaches to quantify the distinction between the NP and AF. This study aimed to discover histogram distribution of the T2 relaxation time of the AF and NP using Gaussian fitting. Our results showed that Gaussian-fitted histogram analysis of T2 relaxation time could achieve quantitative distinction between the NP and AF, and computed Gaussian-fitted histogram parameters had good performance on diagnosing IVDD.
|
The International Society for Magnetic Resonance in Medicine is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.