Oral
Spectroscopy: Neuro
ISMRM & SMRT Annual Meeting • 15-20 May 2021

| Concurrent 2 | 16:00 - 18:00 | Moderators: Mark Brown & Sikandar Shaikh |
 |
0337.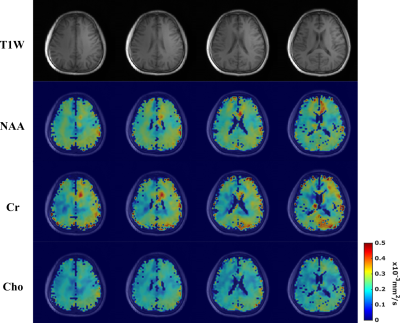 |
High Resolution Volumetric Diffusion-Weighted MRSI Using A Subspace Approach
Zepeng Wang1,2 and Fan Lam1,2
1Department of Bioengineering, University of Illinois Urbana-Champaign, Urbana, IL, United States, 2Beckman Institute for Advanced Science and Technology, Urbana, IL, United States
Diffusion-weighted MRSI (DW-MRSI) promises to significantly expand the capability of in vivo tissue microstructural imaging by simultaneously measuring the diffusion properties of several molecules other than water. However, the applications of DW-MRSI have been mostly limited to either single voxels or 2D experiments with very low resolutions due to several fundamental technical challenges. We describe here a novel method to achieve 3D DW-MRSI with an unprecedented combination of speed, resolution and SNR, by synergizing a special fast sequence and subspace-based processing. We successfully demonstrated high-SNR DW-MRSI of the brain and metabolite-specific ADC maps with the highest ever resolution (3.4×3.4×5.3 mm3).
|
|
0338.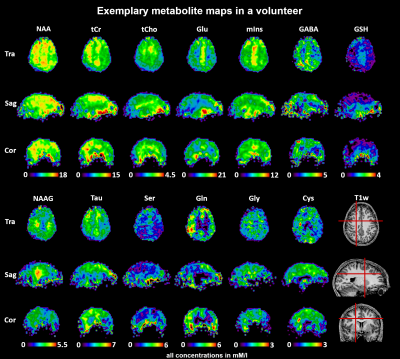 |
3D-CRT-FID-MRSI in the brain at 7T: Evaluation of regional concentration estimates
Gilbert Hangel1,2, Benjamin Spurny3, Philipp Lazen2, Cornelius Cadrien1,2, Sukrit Sharma2, Zoe Käfer2, Nikolaus Doblinger2, Lukas Hingerl2, Eva Hečková2, Bernhard Strasser2, Stanislav Motyka2, Alexandra Lipka2, Stephan Gruber2,
Christoph Brandner4, Rupert Lanzenberger3, Karl Rössler1, Siegfried Trattnig2,5, and Wolfgang Bogner2
1Department of Neurosurgery, Medical University of Vienna, Vienna, Austria, 2High Field MR Centre, Department of Biomedical Imaging and Image-guided Therapy, Medical University of Vienna, Vienna, Austria, 3Division of General Psychiatry, Department of Psychiatry and Psychotherapy, Medical University of Vienna, Vienna, Austria, 4High Field MR Centre, Centre for Medical Physics and Biomedical Engineering, Medical University of Vienna, Vienna, Austria, 5Christian Doppler Laboratory for Clinical Molecular MR Imaging, Vienna, Austria
Applying a 3D-MRSI sequence with ~3 mm isotropic resolution and 15 min measurement time at 7T to a cohort of 24 healthy subjects, we successfully estimated the concentrations of 13 brain metabolites in 44 regions using internal water referencing. We established inter-subject coefficients of variation in the range of 10-20%. The resulting concentration estimates corresponded well to previous research except for GSH.
|
||
 |
0339.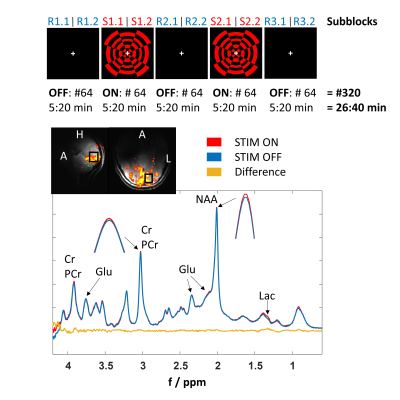 |
Simultaneous Detection of Metabolite Concentration Changes, Water BOLD Signal and pH Changes during Visual Stimulation in the Human Brain at 9.4T
Johanna Dorst1, Tamas Borbath1, and Anke Henning1,2
1High-Field MR Center, Max Planck Institute for Biological Cybernetics, Tübingen, Germany, 2Advanced Imaging Research Center, UT Southwestern Medical Center, Dallas, TX, United States
Previous studies investigated relationships between the BOLD signal and metabolite concentration changes during visual stimulation by sequential or interleaved fMRI/fMRS measurements. The purpose of this study was to simultaneously investigate the dynamics of BOLD signal and metabolite levels in the activated human brain at 9.4T using the metabolite-cycling (MC) technique. A correlation between the MC water dynamics and concentration increases of lactate and glutamate during activation could be verified. Besides, it could be shown that the high spectral quality of fMRS at 9.4T facilitates separate fitting of creatine and phosphocreatine thereby enabling the calculation of pH dynamics during visual stimulation.
|
|
0340.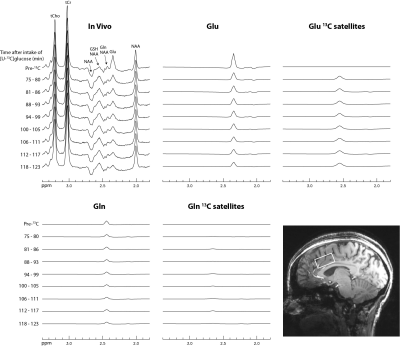 |
In Vivo Measurement of 13C Labeling of Glutamate and Glutamine in the Human Brain Using 1H MRS
Li An1, Shizhe Li1, Maria Ferraris Araneta1, Milalynn Victorino1, Christopher Johnson1, and Jun Shen1
1National Institute of Mental Health, National Institutes of Health, Bethesda, MD, United States
A recently developed single-step spectral editing 1H MRS technique that induces intense Glu and Gln H4 singlets at TE = 56 ms was used to measure fractional enrichments of glutamate and glutamine in the dorsal anterior cingulate cortex (dACC) of five healthy volunteers after oral administration of [U-13C]glucose. This technique offers the ability to measure glutamate neurotransmission in the human brain with the high sensitivity and spatial resolution of 1H MRS using standard commercial equipment.
|
||
 |
0341.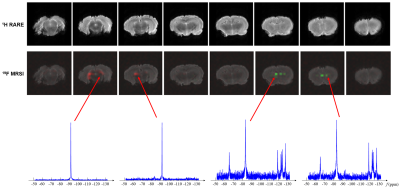 |
Fast High-Resolution 19F-MRSI of Perfluorocarbon Nanoemulsions for MRI Cell Tracking Using SPICE with Learned Subspaces
Yibo Zhao1,2, T. Kevin Hitchens3,4, Michele Herneisey5, Jelena M. Janjic5, Rong Guo1,2, Yudu Li1,2, and Zhi-Pei Liang1,2
1Department of Electrical and Computer Engineering, University of Illinois, Urbana-Champaign, Urbana, IL, United States, 2Beckman Institute for Advanced Science and Technology, University of Illinois, Urbana-Champaign, Urbana, IL, United States, 3Animal Imaging Center, University of Pittsburgh, Pittsburgh, PA, United States, 4Department of Neurobiology, University of Pittsburgh, Pittsburgh, PA, United States, 5Graduate School of Pharmaceutical Sciences, Duquesne University, Pittsburgh, PA, United States
19F-MRSI has the potential to track multiple perfluorocarbon nanoemulsions simultaneously, but existing 19F-MRSI schemes have been limited to CSI, which provides a poor tradeoff between resolution and scan time. In this work, a novel method is proposed to enable fast high-resolution 19F-MRSI. In the proposed method, (k,t)-space is sampled rapidly in EPSI trajectories; data processing is accomplished using a union-of-subspaces model with pre-learned spectral basis. The proposed method has been evaluated using simulation and experimental data, producing encouraging results. The proposed method may open up new opportunities for simultaneous tracking of different labeled cell populations in vivo.
|
|
 |
0342.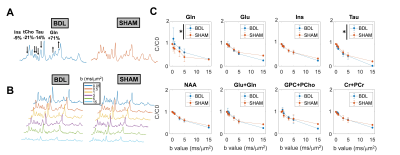 |
Diffusion-weighted magnetic resonance spectroscopy in the cerebellum of a rat model of hepatic encephalopathy at 14.1T
Jessie Mosso1,2,3, Julien Valette4, Katarzyna Pierzchala1,2, Dunja Simicic1,2,3, Ileana Ozana Jelescu1,2, and Cristina Cudalbu1,2
1CIBM Center for Biomedical Imaging, Lausanne, Switzerland, 2Animal Imaging and Technology, EPFL, Lausanne, Switzerland, 3LIFMET, EPFL, Lausanne, Switzerland, 4Commissariat à l'Energie Atomique (CEA), Institut d'Imagerie Biomédicale (I2BM), Molecular Imaging Research Center (MIRCen), Fontenay-aux-Roses, France
Chronic hepatic encephalopathy (cHE) is a severe brain condition arising from chronic liver disease. Microstructural changes occurring due to toxins accumulation in the brain are still unexplored in vivo, especially in cerebellum, and of key interest for disease early detection. Using the STE-LASER sequence, we measured a decreased diffusion coefficient for glutamine and increased for taurine and glutamate in the cerebellum of a rat model of cHE, associated with cell-specific morphological changes measured ex vivo. These preliminary results need to be confirmed by increasing the sample size but they shed light on new aspects in the pathophysiology of HE.
|
|
 |
0343.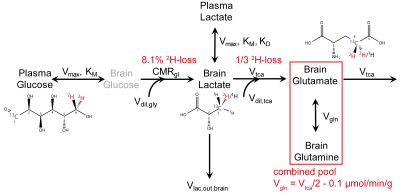 |
Validation of dynamic Deuterium Metabolic Imaging (DMI) for the measurement of cerebral metabolic rates of glucose in rat.
Claudius Sebastian Mathy1,2,3, Monique A. Thomas1, Graeme F. Mason1,4,5, Robin A. de Graaf1,5, and Henk M. De Feyter1
1Department of Radiology and Biomedical Imaging, Magnetic Resonance Research Center, Yale University, New Haven, CT, United States, 2Institute of Physical and Theoretical Chemistry, University of Bonn, Bonn, Germany, 3Department of Diagnostic and Interventional Radiology, RWTH Aachen, Aachen, Germany, 4Department of Psychiatry, Yale University, New Haven, CT, United States, 5Department of Biomedical Engineering, Yale University, New Haven, CT, United States
Deuterium metabolic imaging (DMI) is a novel metabolic imaging technique where 2H MRSI is combined with administration of 2H-labeled substrates. DMI data acquired at metabolic steady-state reveal the relative activities of metabolic pathways, whereas dynamically acquired DMI also provides the metabolic pathway activity rates. The analysis of dynamic DMI data is complicated by the presence of deuterium label loss between deuterated products and water. Here we validated metabolic rates obtained with dynamic DMI with those established using more traditional, 13C-based MR methods.
|
|
 |
0344.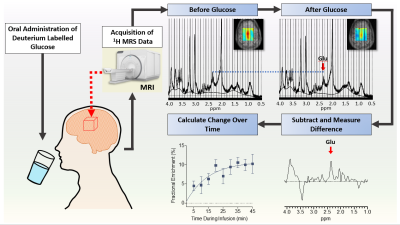 |
Combining 1H MRS and deuterium labeled glucose for mapping of neural metabolism in humans
Abigail T.J. Cember1, Laurie J. Rich1, Puneet Bagga1, Neil E. Wilson2, Ravi Prakash Reddy Nanga1, Deepa Thakuri1, Mark Elliott1, Mitchell D. Schnall3, John A. Detre4, and Ravinder Reddy1
1Center for Magnetic Resonance and Optical Imaging, Department of Radiology, University of Pennsylvania, Philadelphia, PA, United States, 2Siemens Medical Solutions, USA, Malvern, PA, United States, 3Department of Radiology, University of Pennsylvania, Philadelphia, PA, United States, 4Department of Neurology, University of Pennsylvania, Philadelphia, PA, United States
Here we present spectroscopic and chemical shift imaging data from the brains of healthy human subjects acquired using a novel method termed qMRS, which enables tracking of metabolic turnover with the inherent sensitivity of 1H MRS and widespread applicability using standard 1H-based clinical MRI scanners. We demonstrate the feasibility of using qMRS and its corollary chemical shift imaging technique (qCSI) to monitor the temporal and spatial dynamics of metabolite labelling in the human brain following oral consumption of deuterium-labeled glucose. Unlike the related technique of deuterium metabolic imaging (DMI), qMRS does not require implementation of multinuclear MR spectroscopy.
|
|
 |
0345.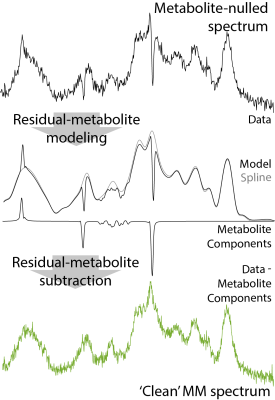 |
The Macromolecular Background Spectrum Does Not Change with Age in Healthy Participants
Steve C.N. Hui1,2, Tao Gong3, Helge J. Zöllner1,2, Yulu Song3, Yufan Chen3, Muhammad G. Saleh4, Mark Mikkelsen1,2, Georg Oeltzschner1,2, Sofie Tapper1,2, Weibo Chen5, Richard A.E. Edden1,2, and Guangbin Wang3
1Russell H. Morgan Department of Radiology and Radiological Science, The Johns Hopkins University School of Medicine, Baltimore, MD, United States, 2F.M. Kirby Research Center for Functional Brain Imaging, Kennedy Krieger Institute, Baltimore, MD, United States, 3Department of Imaging and Nuclear Medicine, Shandong Medical Imaging Research Institute, Cheeloo College of Medicine, Shandong University, Jinan, China, 4Department of Diagnostic Radiology and Nuclear Medicine, University of Maryland School of Medicine, Baltimore, MD, United States, 5Philips Healthcare, Shanghai, China
The aim of this project was to investigate the time course of macromolecular (MM) background spectrum during healthy aging. Recruiting a structured, cross-sectional cohort of 100 participants (10 male and 10 female subject per decade: 20s; 30s; 40s; 50s; and 60s), we acquired metabolite-nulled short-TE PRESS data and modeled the MM spectrum as a series of Gaussian signals at literature-defined chemical shifts. Linear regression of water-scaled MM signal areas revealed no significant relationship between age and MM signal areas, suggesting the MM spectrum may be more stable than has been suggested in the literature.
|
|
0346.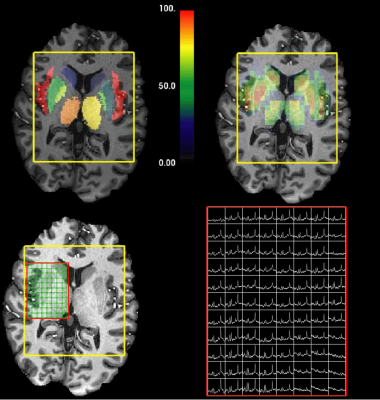 |
Ultra high-field, high-resolution, multi-voxel MRS in pre-manifest and early-manifest Huntington's Disease
Yan Li1, Huawei Liu1, Angela Jakary1, Sivakami Avadiappan1, Melanie Morrison1, Ralph Noeske2, Peder E.Z. Larson1, Alexandra Nelson3, Katherine Possin3, Michael Geschwind3, Christopher Hess1, and Janine M Lupo1
1Department of Radiology and Biomedical Imaging, University of California San Francisco, San Francisco, CA, United States, 2GE Healthcare, Munich, Germany, 3Department of Neurology, University of California San Francisco, San Francisco, CA, United States
In this study of Huntington's Disease (HD), we evaluated brain metabolites from pre-manifest HD (PM) and early-manifest HD (EM), as well as their relationship to disease burden and motor disturbances. We used multi-voxel MRS at ultra-high field strength (7T), in order to improve the reliability and sensitivity of detecting focal metabolic changes that are related to HD. We found metabolic alterations in the thalamus and insula and changes in choline, mI, glutathione and glutamate that were correlated with clinical measures of disease severity.
|
The International Society for Magnetic Resonance in Medicine is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.