Oral
fMRI of Animal Models
ISMRM & SMRT Annual Meeting • 15-20 May 2021

| Concurrent 3 | 14:00 - 16:00 | Moderators: Giovanna Diletta Ielacqua & Jin Lee |
 |
0509.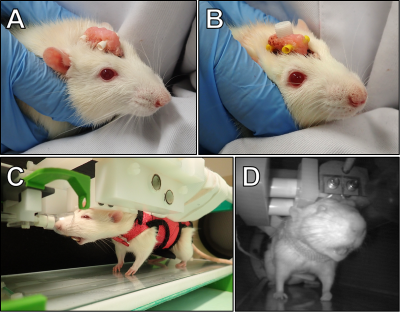 |
MB-SWIFT fMRI studies in head-fixed behaving rats
Jaakko Paasonen1, Petteri Stenroos1,2, Hanne Laakso1, Tiina Pirttimäki1, Ekaterina Zhurakovskaya1, Raimo A Salo1, Heikki Tanila1, Djaudat Idiyatullin3, Michael Garwood3, Shalom Michaeli3, Silvia Mangia3, and Olli Gröhn1
1A.I.V. Institute for Molecular Sciences, University of Eastern Finland, Kuopio, Finland, 2Grenoble Institut des Neurosciences, Grenoble, France, 3Center for Magnetic Resonance Research, University of Minnesota, Minneapolis, MN, United States
Currently there are no tools to study simultaneously whole-brain processing and behavior in rats. Here we introduce a novel approach for fMRI studies in head-fixed and minimally restrained rats that can express behavior. This was achieved with MB-SWIFT sequence that is both quiet and insensitive to movement. First, the whole brain, including hindbrain, was functionally parcellated with high fidelity. Second, fMRI maps showing activation in relevant networks during spontaneous single behavioral events were successfully generated. Our approach links global network activity to behavior and has potential to enable novel experimental designs in neuroscience studies.
|
|
0510.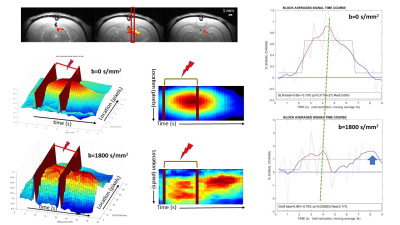 |
Linescan BOLD and diffusion fMRI signal responses triggered by activation of the rat visual system differ in time, amplitude and shape
Denis Le Bihan1, Luisa Ciobanu1, Yukiko Masaki1,2, and Erwan Selingue1
1NeuroSpin, Gif-sur-Yvette, France, 2Shionogi & Co., Ltd., Osaka, Japan
BOLD and diffusion fMRI signal response time courses following rat visual stimulation differ in time, amplitude and shape.
|
||
0511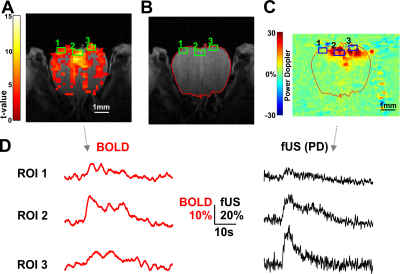 |
BOLD fMRI and functional ultrasound comparisons in co-registered olfactory bulb sections of the same mice Video Permission Withheld
Davide Boido1, Ali-Kémal Aydin2, Yannick Goulam Houssen2, Demené Charlie3, Mickael Tanter3, Serge Charpak2, and Luisa Ciobanu1
1NeuroSpin, CEA, Paris, France, 2Inserm - Institut de la Vision, Paris, France, 3Physics for Medicine, ESPCI, INSERM, CNRS, PSL Research University, Paris, France Using UHF (17.2T) BOLD fMRI on the mouse Olfactory Bulb (OB) we recorded odor activation at high spatio-temporal resolution (0.17x0.17x0.3 mm; TR= 250ms). With 6% 1s odor application we found fast unimodal activations. Increasing odor concentration and duration (35% 5s) we revealed the second vascular component. Adding functional ultrasound (fUS) recordings from the co-registered OB coronal section, we could verify an overall good accordance in the dynamics of BOLD and fUS signals in small ROIs (< 0.5 mm^3), although evident differences were also found in some ROIs. We aim at highlighting the relationship between local vascular activity and BOLD fMRI. |
||
 |
0512.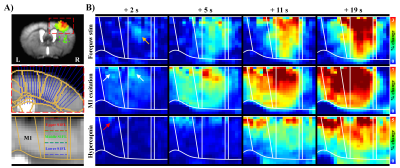 |
Neural activity-driven BOLD responses within the cortex occur first at synaptic input layers
Won Beom Jung1, Geun Ho Im1, Haiyan Jiang1,2, and Seong-Gi Kim1,2
1Center for Neuroscience Imaging Research (CNIR), Institute for Basic Science (IBS), Suwon, Korea, Republic of, 2Department of Biomedical Engineering, Sungkyunkwan University, Suwon, Korea, Republic of
To further advance understanding of brain functions, identifying the direction of information flow, such as thalamocortical vs. corticothalamic projections, is critical. Because the early hemodynamic response at microvessels near active neurons can be detected by ultrahigh field fMRI, we propose using the onset times of fMRI responses to discern the information flow. This approach was confirmed by observing the high temporal resolution BOLD fMRI responses to bottom-up somatosensory stimulation and top-down optogenetic stimulation of the primary motor cortex in anesthetized mice at ultrahigh field of 15.2 T.
|
|
0513.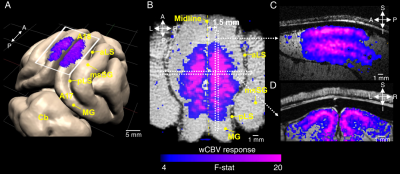 |
Layer-specific orientation selectivity in cat visual cortex using 9.4 Tesla fMRI and multi-photon optical imaging
Shinho Cho1, Arani Roy2, Chao Liu2, Djaudat Idiyatullin1, Wei Zhu1, Yi Zhang1, Xiao-Hong Zhu1, Prakash Kara2, Wei Chen1, and Kâmil Uğurbil1
1Center for Magnetic Resonance Research and Department of Radiology, University of Minnesota, Minneapolis, MN, United States, 2Center for Magnetic Resonance Research and Department of Neuroscience, University of Minnesota, Minneapolis, MN, United States
Using high isotropic resolution (250 μm), cerebral blood volume weighted (wCBV) fMRI, we examined whether layer-specific cortical signals could be detected upon visual stimulation in cat primary visual cortex. We also examined single blood vessel responses (dilation, blood flow) to identical stimuli by using 2- and 3-photon imaging in the same cortical area and species. With fMRI, we often found orientation preference maps tangential and orthogonal to the cortical surface. Moreover, the laminar profile of orientation selectivity with both imaging techniques (fMRI and optical) revealed a selectivity index that was significantly lower in cortical layer 4 compared to layer 2/3.
|
||
 |
0514.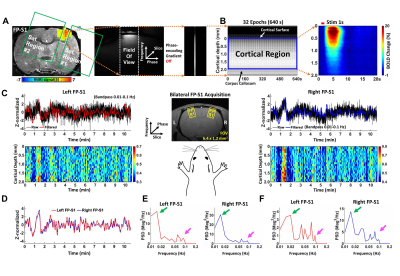 |
Characterize laminar-specific interhemispheric functional coherence in resting-state fMRI using bilateral line-scanning fMRI (BiLS)
Sangcheon Choi1,2, Yi Chen1, Hang Zeng1,2, and Xin Yu1,3
1Max Planck Institute for Biological Cybernetics, Tuebingen, Germany, 2Graduate Training Centre of Neuroscience, Tuebingen, Germany, 3MGH/MIT/HMS Athinoula A. Martinos Center for Biomedical Imaging, Department of Radiology, Harvard Medical School, Massachusetts General Hospital, Charlestown, MA, United States
We developed a bilateral line-scanning fMRI method to investigate interhemispheric slow fluctuations (< 0.1 Hz) with laminar specificity in resting-state fMRI in anesthetized rats. Based on the coherence analysis, two distinct slow fluctuation features in symmetric cortices were identified: ultra-slow fluctuation (0.01-0.02 Hz) was synchronized across all cortical laminae, and Layer 2/3 specific slow fluctuations (0.08-0.1 Hz). In contrast to the ultra-slow fluctuation related to global brain state changes, the Layer 2/3 specific slow fluctuation is more likely associated with intrinsic neuronal correlation driven by the callosal projection.
|
|
 |
0515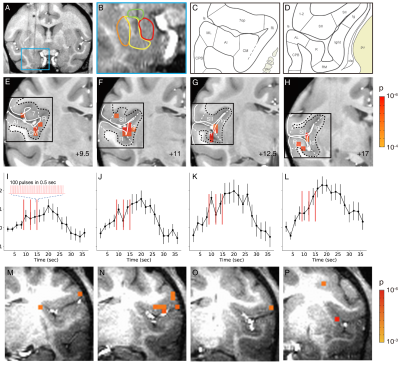 |
A rapid in vivo method for mapping cortical connections of primate amygdala with infrared neural stimulation and 7T fMRI Video Permission Withheld
Augix Guohua Xu1, Sunhang Shi1, Yunyun Rui1, Xiaotong Zhang1, Lizabeth Romanski2, Katalin M. Gothard3, and Anna Wang Roe1
1Interdisciplinary Institute of Neuroscience and Technology, Zhejiang University, Hangzhou, China, 2Dept of Neuroscience, University of Rochester School of Medicine, Rochester, NY, United States, 3University of Arizona, Dept of Physiology, Tucson, AZ, United States
We have previously shown that INS-fMRI is a rapid method for mapping mesoscale brain networks in the macaque monkey brain. Here, we extend this capability by stimulating deep brain sites. We test this new method by stimulating the basal nucleus of amygdala in the macaque monkey. The connections we identified are consistent with dye-tracing studies. In conclusion, our results indicate that INS-fMRI is a promising novel method for mapping connections of deep brain structures at high spatial resolution.
|
|
 |
0516.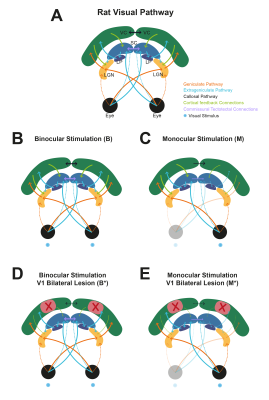 |
Dissecting the impact of cortical feedback and inhibitory tectotectal loops in negative BOLD responses along the rat visual pathway
Rita Gil1, Mafalda Valente1, Alfonso Renart1, and Noam Shemesh1
1Champalimaud Centre for the Unknown, Lisbon, Portugal
The underlying source of negative BOLD responses (NBRs) is still debated. Here, we show that binocular visual stimulation with short inter-stimulus-intervals (ISIs) lead to strong NBRs in the rat superior colliculus (SC). We perturbed cortical feedback loops with V1 ibotenic acid lesions and input to the ipsilateral SC via tectotectal projections through monocular stimulation. SC NBRs were upward regulated upon both V1 lesioning and monocular stimulation of non-lesioned animals. When combining reduced cortical feedback and tectotectal projections, NBRs in SC were abolished. Our results suggest an important role for corticotectal and commissural tectotectal projections in SC NBRs at short ISIs.
|
|
0517.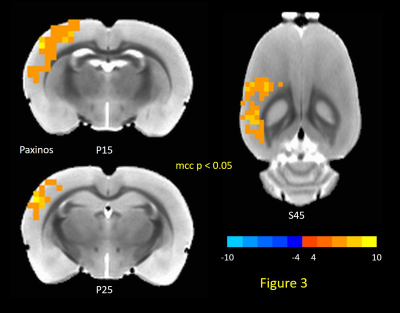 |
Investigating Neurophysiological Basis of Resting State fMRI Signal Components through Suppression of Cortical Slow Rhythms
Vahid Khalilzad Sharghi1, Eric Maltbie1, Wen-Ju Pan1, Shella Keilholz1, and Kaundinya Gopinath2
1Biomedical Engineering, Emory University/Georgia Institute of Technology, Atlanta, GA, United States, 2Department of Radiology & Imaging Sciences, Emory University, Atlanta, GA, United States
In this study, we tested hypothesis advanced by some groups that brain slow rhythms serve as the neurophysiological basis of resting state fMRI (rsfMRI). Putative suppression of cortical rhythms with an established technique, led to significant reduction in the amplitude of rsfMRI quasi-periodic patterns (QPPs), and enhancement in the rsfMRI measures of intrinsic functional connectivity FC in canonical brain function networks in rats. The results indicate cortical slow rhythms serve as the genesis of only the vigilance dependent components (e.g., QPP) of rsfMRI signals. Further attenuation of these non-specific signals enhances delineation of brain function networks.
|
||
 |
0518.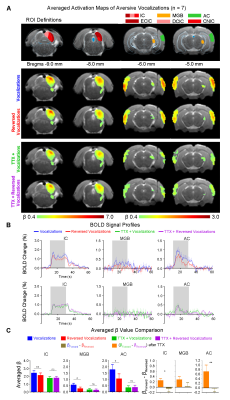 |
Pharmacological inactivation of ventral hippocampus disrupts central auditory processing
Eddie C. Wong1,2, Xunda Wang1,2, Vick Lau1,2, Alex T.L. Leong1,2, and Ed X. Wu1,2
1Laboratory of Biomedical Imaging and Signal Processing, The University of Hong Kong, Hong Kong SAR, China, 2Department of Electrical and Electronic Engineering, The University of Hong Kong, Hong Kong SAR, China
Audition is vital for communication, learning and memory processes. However, the hippocampus, which can support these functions, is absent from networks of auditory processing. To bridge this gap, we employed auditory fMRI and pharmacological inactivation techniques to directly characterize how hippocampal outputs affect auditory responses to auditory stimuli in primary auditory-associated structures. Using behaviorally-relevant, natural sounds for rodent behaviors, or their temporally-reversed counterparts, we revealed that absence of hippocampal output disrupts auditory responses to vocalizations in auditory midbrain, thalamus and cortex. For the first time, our results demonstrated the critical role of hippocampus in shaping response selectivity to behaviorally-relevant sounds.
|
The International Society for Magnetic Resonance in Medicine is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.