Georgios Batsios1, Celine Taglang1, Anne Marie Gillespie1, Peder Larson1, Sabrina M Ronen1, and Pavithra Viswanath1
1Radiology and Biomedical Imaging, UCSF, San Francisco, CA, United States
1Radiology and Biomedical Imaging, UCSF, San Francisco, CA, United States
We
show that assessment of glucose metabolism via the pentose phosphate pathway using
hyperpolarized δ-[1-13C]gluconolactone informs on early response to
chemotherapy in preclinical glioma models. Our results have the potential to
enhance therapy assessment for glioma patients.
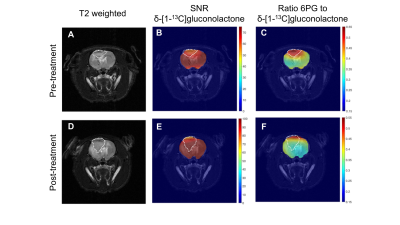
Hyperpolarized
δ-[1-13C]gluconolactone
informs on early response to TMZ treatment in vivo. Top: representative
metabolic maps from a BT88-bearing rat pre-treatment
with TMZ. T2 weighted MRI (A),
heatmap of SNR of hyperpolarized δ-[1-13C]gluconolactone (B) and heatmap of 6PG/δ-[1-13C]gluconolactone ratio (C). Bottom: metabolic maps from a BT88-bearing rat post-treatment with TMZ. T2 weighted MRI (D), heatmap of SNR
of hyperpolarized δ-[1-13C]gluconolactone
(E) and
heatmap of 6PG/δ-[1-13C]gluconolactone
ratio (F). Tumor is delineated in white.
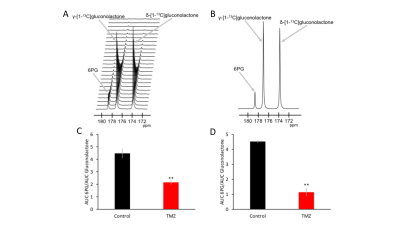
Hyperpolarized δ-[1-13C]gluconolactone
can monitor response to temozolomide (TMZ) in glioma cells. Representative 13C-MRS spectral
array (A) and summed 13C spectra (B) showing 6PG production from
hyperpolarized δ-[1-13C]gluconolactone
in live U87 cells. Effect of TMZ on 6PG production in U87 (C) and GS2 (D) cells (N=3).